## Clinical Context: High-Risk B-ALL in a Child This patient has: - **B-cell ALL** confirmed on bone marrow - **High-risk features:** Age 6 years (borderline), WBC 120,000/μL (>50,000/μL threshold) - **Hemodynamic stability** — no hyperleukostasis or tumor lysis syndrome - **No CNS involvement** on clinical examination ## Why Induction Chemotherapy After Baseline Workup is Correct **Key Point:** Standard-risk and high-risk B-ALL require immediate multi-agent induction chemotherapy (VDAD regimen) after establishing IV access and confirming baseline organ function and uric acid levels. **High-Yield:** The VDAD induction regimen (Vincristine, Daunorubicin, Asparaginase, Dexamethasone) is the gold standard for childhood ALL and achieves complete remission in >95% of children when combined with risk-adapted intensification. **Clinical Pearl:** Delay in starting chemotherapy in ALL is associated with worse outcomes. Baseline investigations (renal function, uric acid, LDH, phosphate) must be obtained rapidly (within hours) to assess tumor lysis risk, but should NOT delay induction initiation. ### Pre-Induction Workup (Parallel, Not Sequential) ```mermaid flowchart TD A[B-ALL diagnosis confirmed]:::outcome --> B[Establish central venous access]:::action B --> C[Baseline labs in parallel]:::action C --> D[Renal function, uric acid, LDH, phosphate]:::action D --> E[Assess tumor lysis risk]:::decision E -->|Low risk| F[Start VDAD induction]:::action E -->|High risk| G[Allopurinol + hydration first]:::action G --> F F --> H[Intrathecal MTX during induction]:::action H --> I[Consolidation & intensification]:::action ``` **Mnemonic:** **VDAD = Vincristine, Daunorubicin, Asparaginase, Dexamethasone** (standard induction) ## Why NOT the Other Options? | Approach | Rationale for Rejection | |----------|------------------------| | Hydroxyurea + allopurinol alone, then refer | Delays definitive chemotherapy. Hydroxyurea is not curative in ALL and 1-week delay increases relapse risk. | | Immediate intrathecal MTX before induction | ITM is given DURING induction (weeks 1–5), not before. CNS prophylaxis is part of the protocol, not the first step. | | Imatinib as first-line | Imatinib is reserved for BCR-ABL+ ALL (Philadelphia chromosome), which is NOT mentioned here. Standard B-ALL is imatinib-resistant. | **Warning:** Delaying chemotherapy in childhood ALL by even 1–2 weeks increases relapse risk and worsens survival. Baseline investigations must be done in parallel with IV access, not sequentially. ## Tumor Lysis Syndrome Prophylaxis **High-Yield:** This patient has high tumor burden (WBC 120,000/μL) but is hemodynamically stable. Prophylaxis is still needed: 1. **Allopurinol** 10 mg/kg/day (or febuxostat in high-risk) 2. **Aggressive hydration** (2–3 L/m²/day) to maintain urine output >200 mL/m²/h 3. **Avoid diuretics** unless fluid overload 4. **Monitor K^+^, PO₄^3−^, uric acid** q6–12h during first 48 h 5. **Rasburicase** if uric acid >8 mg/dL or rapid rise **Clinical Pearl:** Intrathecal methotrexate is given as part of induction (typically weeks 1, 3, 5) and continuation therapy, not before induction. CNS-directed therapy is essential in ALL because the blood–brain barrier excludes systemic chemotherapy. [cite:Robbins 10e Ch 13; Harrison 21e Ch 110] 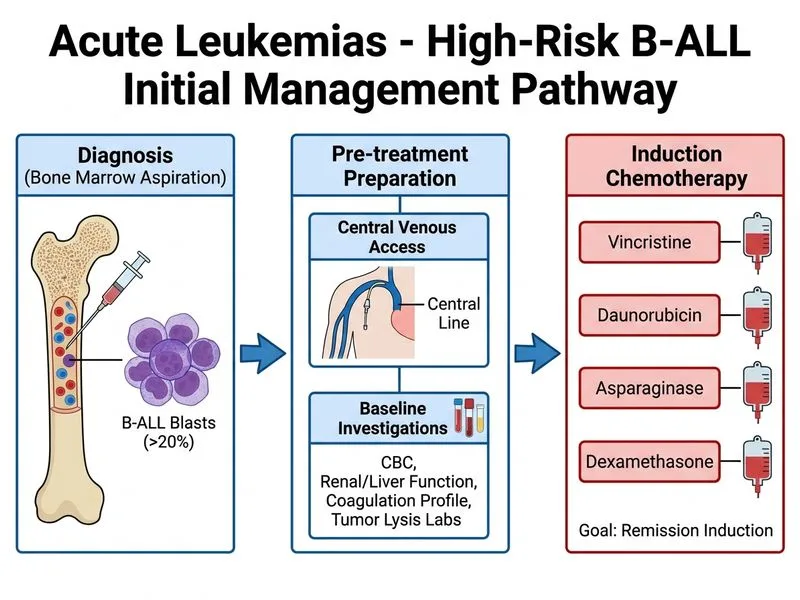
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.