## Extrinsic Apoptotic Pathway — FADD and the DISC **Key Point:** FADD (Fas-Associated protein with Death Domain) is the critical adaptor protein that directly recruits and activates caspase-8 at the death receptor, forming the Death-Inducing Signaling Complex (DISC). ### Sequential Assembly of the Extrinsic Pathway 1. **Death Receptor Ligation**: Extracellular ligands (FasL, TNF-α, TRAIL) bind to death receptors (Fas, TNF-R1, TRAIL-R). 2. **Receptor Trimerization & Clustering**: Ligand binding induces receptor aggregation at the plasma membrane. 3. **FADD Recruitment**: The death domain (DD) of the receptor recruits FADD via homotypic DD–DD interactions. 4. **Caspase-8 Binding**: FADD's death effector domain (DED) recruits procaspase-8 (also via DED–DED interactions). 5. **DISC Formation**: The assembled complex (receptor + FADD + procaspase-8) is called the Death-Inducing Signaling Complex (DISC). 6. **Caspase-8 Activation**: Procaspase-8 undergoes autoproteolytic activation at the DISC. 7. **Caspase Cascade**: Active caspase-8 directly activates executioner caspases (caspase-3/7) or can amplify the signal via the intrinsic pathway (caspase-8 → BID → mitochondria). **High-Yield:** FADD is the **direct linker** between the death receptor and caspase-8. Without FADD, the extrinsic pathway cannot initiate. ### Intrinsic vs. Extrinsic Pathway — Adaptor Proteins | Pathway | Primary Sensor | Adaptor Protein | Initiator Caspase | Apoptosome | |---------|---|---|---|---| | **Intrinsic** | p53 (stress) | None (BCL-2 family) | Caspase-9 | Yes (Apaf-1 + Cyt c) | | **Extrinsic** | Death receptor | FADD | Caspase-8 | No | **Clinical Pearl:** Mutations in FADD or caspase-8 cause ALPS (Autoimmune Lymphoproliferative Syndrome), characterized by impaired lymphocyte apoptosis and autoimmunity. **Mnemonic:** **"FADD = Fas-Associated Death Domain"** — it is the bridge between Fas (death receptor) and caspase-8. 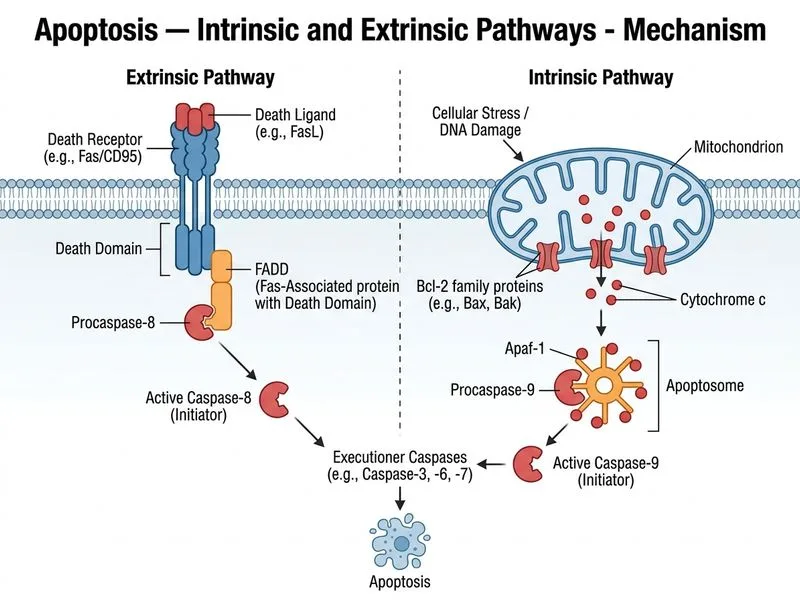
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.