## Clinical Severity Assessment This patient meets criteria for **severe, refractory atopic dermatitis**: - **Extent:** >50% BSA involvement (very extensive) - **Duration of inadequate response:** 6 months on high-potency topical corticosteroids - **Impact:** Significant sleep disturbance, functional impairment (work absence) - **Baseline labs:** Normal renal and hepatic function (prerequisite for systemic agents) ## Systemic Therapy Hierarchy in Severe AD ```mermaid flowchart TD A["Severe AD unresponsive to topical therapy"]:::outcome --> B{"Baseline labs normal?"}:::decision B -->|No| C["Optimize labs, treat comorbidities"]:::action B -->|Yes| D{"Dupilumab available/affordable?"}:::decision D -->|Yes| E["Dupilumab: First-line biologic"]:::action D -->|No/Contraindicated| F{"Active infection or malignancy?"}:::decision F -->|Yes| G["Treat infection/malignancy first"]:::urgent F -->|No| H["Cyclosporine (alternative biologic unavailable)"]:::action E --> I["Assess response at 4 weeks"]:::outcome H --> I ``` ## Dupilumab: Why It Is the Best Next Step | Feature | Dupilumab | Cyclosporine | |---------|-----------|---------------| | **Mechanism** | Anti-IL-4Rα (blocks IL-4 and IL-13) | Non-selective T-cell immunosuppressant | | **Efficacy in severe AD** | 75–90% achieve EASI-75 at 16 weeks | 60–70% response rate | | **Safety profile** | Well-tolerated; no organ toxicity monitoring | Requires renal/hepatic monitoring; teratogenic | | **Long-term use** | Safe for >2 years | Limited to 3–4 months due to cumulative toxicity | | **First-line biologic** | **YES** (FDA, EMA, Indian guidelines 2023) | Second-line (if dupilumab unavailable/contraindicated) | | **Pregnancy** | Compatible with pregnancy | Contraindicated | | **Cost** | Higher initial cost, but long-term benefit | Lower cost, but limited duration | **Key Point:** Dupilumab is the current gold-standard first-line biologic for severe atopic dermatitis that has failed topical therapy. It targets the IL-4 receptor, blocking both IL-4 and IL-13 signaling—the central drivers of Th2 inflammation in AD. **High-Yield:** The 2023 Indian Academy of Dermatology and WHO guidelines recommend dupilumab as the preferred systemic biologic for severe AD. Baseline IgE and eosinophil count are measured to document baseline atopy severity and monitor response; they are NOT contraindications to treatment. **Clinical Pearl:** Dupilumab typically shows clinical improvement within 2–4 weeks (faster than cyclosporine). The initial loading dose is 600 mg (two 300 mg injections), followed by 300 mg every 2 weeks. No organ toxicity monitoring is required, making it safer for long-term use than cyclosporine. **Mnemonic:** **DUPILUMAB = DUal Pathway IL-4 Receptor** (blocks both IL-4 and IL-13 via shared IL-4Rα chain). 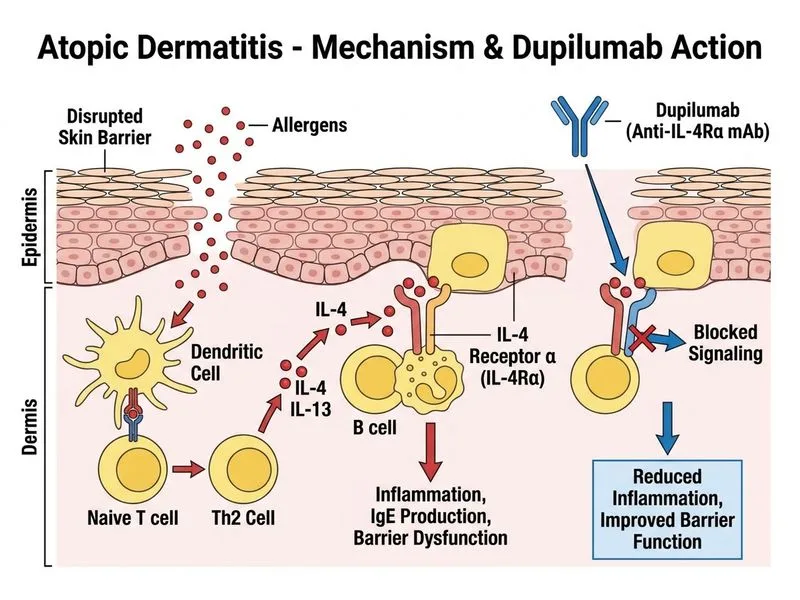
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.