## Severe Atopic Dermatitis: Systemic and Biologic Therapy ### Clinical Assessment This patient has **severe atopic dermatitis** characterized by: - Extensive involvement: >60% body surface area (BSA) - Severe symptoms: widespread erythema and exudation - Treatment failure: inadequate response to potent topical corticosteroids - Recurrent secondary infections (6 months of repeated antibiotic courses) - Markedly elevated serum IgE (2400 IU/mL, indicating strong Th2 bias) - Long disease duration (10 years) **Key Point:** When topical therapy fails and BSA involvement is >10%, systemic therapy is indicated [cite:Inamadar & Palit; IAD Guidelines]. ### Systemic and Biologic Options for Severe AD | Agent | Mechanism | Efficacy | Safety Profile | First-Line Status | |-------|-----------|----------|-----------------|-------------------| | **Dupilumab** | Anti-IL-4Rα (blocks IL-4 and IL-13) | Excellent (EASI-75 ~70%) | Well-tolerated; monitor for conjunctivitis | **YES — preferred** | | Cyclosporine | Calcineurin inhibitor; T-cell suppression | Good (EASI-75 ~50%) | Nephrotoxicity, hypertension, infection risk | Second-line | | Methotrexate | Antimetabolite; immunosuppression | Moderate | Hepatotoxicity, bone marrow suppression | Third-line | | Systemic corticosteroids | Broad immunosuppression | Rapid but temporary | Rebound flares, long-term toxicity | **NOT for maintenance** | **High-Yield:** Dupilumab is now the preferred biologic for moderate-to-severe AD in adults and children ≥6 years. It targets the IL-4 receptor alpha subunit, blocking both IL-4 (Th2) and IL-13 (Th2/Th22) signaling — the core pathogenic axis in AD [cite:Harrison 21e Ch 309; FDA approval 2017]. ### Why Dupilumab Is Optimal Here 1. **Superior efficacy:** EASI-75 response rates ~70% in pivotal trials (SOLO 1 & 2) 2. **Rapid onset:** Clinical improvement within 2 weeks 3. **Sustained remission:** Maintains control with continued dosing 4. **Safety:** No nephrotoxicity, no hepatotoxicity, no bone marrow suppression 5. **Addresses pathophysiology:** Directly targets the IL-4/IL-13 axis driving Th2 inflammation in AD 6. **Indication met:** Approved for moderate-to-severe AD with inadequate response to topical therapy **Clinical Pearl:** Dupilumab may cause conjunctivitis (10–15% of patients) — counsel the patient and monitor. This is reversible and rarely requires discontinuation. ### Why Other Agents Are Suboptimal **Cyclosporine (Option A):** - Second-line agent (not first-line biologic) - Requires baseline and ongoing renal function monitoring - Risk of hypertension and infection - Less effective than dupilumab (EASI-75 ~50%) - Typically reserved if dupilumab is contraindicated or unavailable **Methotrexate (Option C):** - Third-line systemic agent - Slower onset (weeks to months) - Requires monitoring for hepatotoxicity and bone marrow suppression - Less effective than dupilumab or cyclosporine for AD - Better suited for psoriasis **Systemic Corticosteroids (Option D):** - **Warning:** NOT recommended for maintenance therapy in AD - Rapid onset but causes rebound flares upon withdrawal - Long-term use risks: immunosuppression, infection, osteoporosis, metabolic effects - May be used briefly (1–2 weeks) during acute flares only, not for 4-week courses - This patient's recurrent infections make systemic steroids contraindicated **Mnemonic:** **BIDS** for biologic therapy in severe AD = **B**iologic (dupilumab first), **I**nadequate topical response, **D**isease >10% BSA, **S**evere symptoms/impact on quality of life. ### Dosing and Monitoring for Dupilumab - Loading dose: 600 mg SC (or 400 mg if <60 kg) - Maintenance: 300 mg SC every 2 weeks - Baseline assessment: CBC, liver function, IgE level (optional) - Monitor: Clinical response (EASI, pruritus NRS) at weeks 2, 4, 12 - Counsel: Conjunctivitis risk, injection technique, adherence 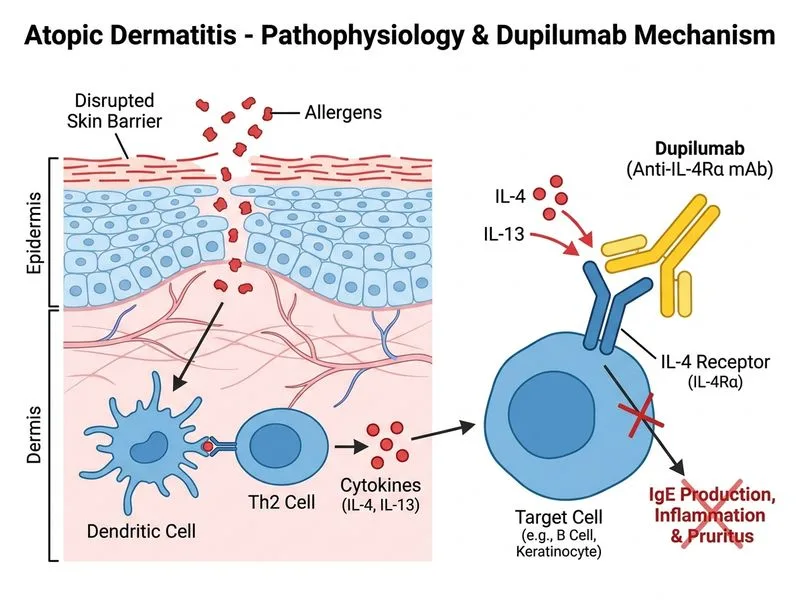
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.