## Investigation Choice: Distant Staging in Node-Positive Breast Cancer ### Clinical Context The patient has node-positive breast cancer (pN1: 1/2 sentinel nodes involved, 0/12 additional nodes = pN1a) requiring accurate distant staging (M-staging) before adjuvant systemic therapy planning. ### Why CT Chest, Abdomen, and Pelvis is Correct **Key Point:** CT chest, abdomen, and pelvis (CAP) is the standard investigation for M-staging in node-positive breast cancer. It detects visceral, hepatic, and pelvic metastases with high sensitivity and specificity [cite:NCCN Breast Cancer Guidelines 2023]. **High-Yield:** CT CAP is recommended for all patients with: - Node-positive disease (pN1, pN2, pN3) - Locally advanced breast cancer (T3, T4) - Symptoms suggestive of metastatic disease **Clinical Pearl:** CT CAP has ~85–90% sensitivity for detecting metastatic disease in the lungs, liver, and peritoneal cavity. It is more sensitive than chest X-ray alone and does not expose the patient to the radiation burden of PET-CT in asymptomatic node-positive disease. ### Comparison of Distant Staging Investigations | Investigation | Sensitivity | Specificity | Indication | Radiation Burden | Cost | |---|---|---|---|---|---| | **CT CAP** | 85–90% | 90% | Standard for pN+ disease | Moderate (CT) | Moderate | | **PET-CT** | 90–95% | 85% | Symptomatic or high-risk disease | High (CT + PET) | High | | **Chest X-ray + LFTs** | 60–70% | 80% | Initial screening (outdated) | Low | Low | | **Bone scan** | 70–80% | 85% | Symptomatic bone pain (outdated) | Moderate | Low | **Mnemonic: CAP for Node-Positive** — CT Abdomen and Pelvis: standard for Axillary node-positive breast cancer, Practical, cost-effective. ### Staging Algorithm for Node-Positive Breast Cancer ```mermaid flowchart TD A[Invasive breast cancer with pN1/pN2/pN3]:::outcome --> B{Symptoms of metastatic disease?}:::decision B -->|No symptoms| C[CT chest, abdomen, pelvis]:::action B -->|Bone pain, neurologic symptoms| D[Add bone scan or PET-CT]:::action C --> E{Metastases detected?}:::decision E -->|No M0| F[Adjuvant chemotherapy + endocrine/targeted therapy]:::action E -->|Yes M1| G[Palliative systemic therapy, clinical trial consideration]:::action D --> H[Complete staging, treatment planning]:::outcome ``` ### When Each Investigation is Used **CT CAP (Standard):** - Node-positive disease (pN1–pN3) - Locally advanced disease (T3–T4) - Asymptomatic staging **PET-CT (Selective):** - High-risk disease (pN3, T4) - Symptoms suggestive of metastases - Equivocal findings on CT - Not routinely recommended for asymptomatic pN1 disease due to cost and radiation **Bone Scan (Outdated):** - Largely replaced by PET-CT - Low specificity (high false-positive rate from benign bone disease) - Reserved for symptomatic bone pain if PET-CT unavailable **Chest X-ray + LFTs (Insufficient):** - Inadequate sensitivity for detecting small metastases - No longer recommended as sole staging modality for node-positive disease - May be used as initial screening in resource-limited settings 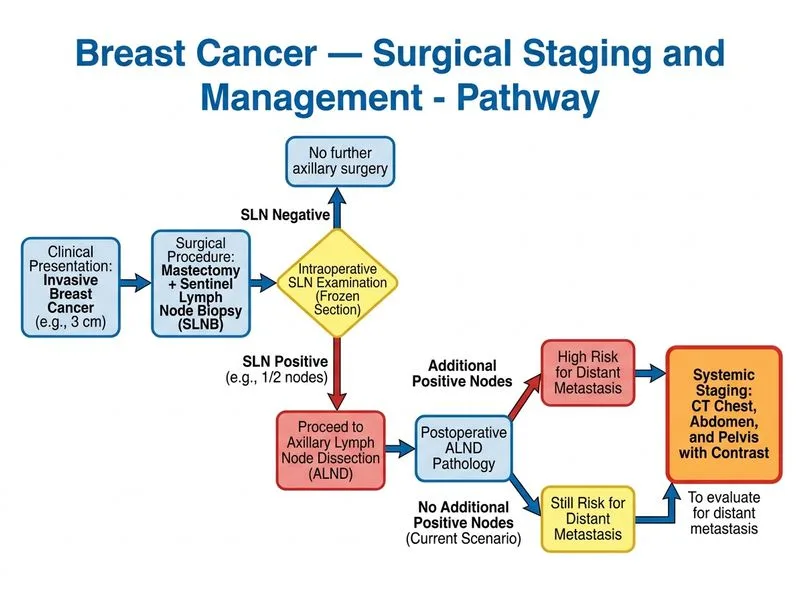
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.