## TNM Staging ### Component Assessment | Component | Finding | Stage | |-----------|---------|-------| | Tumour size | 3.5 cm | T2 | | Axillary nodes | 4 of 12 involved | N2a (4–9 nodes) | | Distant metastases | None | M0 | | **Overall Stage** | **T2N2aM0** | **IIIA** | **Key Point:** Stage IIIA is defined as any T with N2 (4–9 positive nodes) and M0, or T3 with N1–N2 and M0. This patient has T2N2aM0, which is stage IIIA (locally advanced disease). ## Adjuvant Therapy Rationale ### Chemotherapy Indication **High-Yield:** Locally advanced breast cancer (stage IIIA) with nodal involvement mandates systemic chemotherapy regardless of hormone receptor status. The presence of 4 involved nodes confers high risk of micrometastatic disease. **Regimen: AC-T (Doxorubicin–Cyclophosphamide followed by Taxane)** - Standard for node-positive and high-risk node-negative disease - Improves disease-free and overall survival in stage II–III cancers - Typical schedule: AC × 4 cycles → Paclitaxel × 12 weeks (or docetaxel × 4 cycles) ### Endocrine Therapy **Clinical Pearl:** ER/PR-positive tumours require long-term tamoxifen (5 years) or aromatase inhibitors (AI) in postmenopausal women. This patient is postmenopausal with ER/PR-positive disease. - **Tamoxifen** is appropriate and cost-effective in India - Alternatively, an AI (letrozole, anastrozole) may be used - Therapy begins after completion of chemotherapy ### HER2 Status **Warning:** HER2 is **negative** in this case. Trastuzumab (anti-HER2 monoclonal antibody) is indicated ONLY for HER2-positive tumours. Do not confuse HER2-negative with HER2-positive disease. ## Treatment Algorithm ```mermaid flowchart TD A[Stage IIIA Breast Cancer<br/>T2N2aM0]:::outcome --> B{Chemotherapy<br/>indicated?}:::decision B -->|Yes<br/>Node-positive| C[AC-T regimen<br/>4 + 12 weeks]:::action C --> D{ER/PR status?}:::decision D -->|Positive| E[Tamoxifen 5 years]:::action D -->|Negative| F[No endocrine therapy]:::outcome G{HER2 status?}:::decision D -->|Check| G G -->|Positive| H[Add Trastuzumab<br/>1 year]:::action G -->|Negative| I[No anti-HER2 therapy]:::outcome E --> J[Follow-up imaging<br/>& surveillance]:::action ``` **Mnemonic: Adjuvant Therapy Selection — "CHERT"** - **C**hemotherapy — node-positive, high-grade, large tumours - **H**ormone therapy — ER/PR-positive (tamoxifen or AI) - **E**rB2 (HER2) therapy — HER2-positive only (trastuzumab) - **R**adiation — post-mastectomy if ≥4 nodes or T4 disease - **T**arget therapy — other molecular subtypes (CDK4/6i, etc.) ## Why Not the Other Options **Option 1 (Correct):** Stage IIIA with AC-T + tamoxifen matches the TNM stage and receptor profile. **Option 2 (Tamoxifen alone):** Tamoxifen monotherapy is inadequate for stage IIIA disease. Chemotherapy is mandatory for node-positive, locally advanced cancers to reduce recurrence and improve survival. **Option 3 (Trastuzumab):** HER2 is negative. Trastuzumab is contraindicated and offers no benefit in HER2-negative tumours. Adding it would increase toxicity without therapeutic gain. **Option 4 (Stage IIB):** Misclassification. T2N2aM0 is stage IIIA, not IIB. Stage IIB is T3N0 or T2N1. The presence of 4 involved nodes (N2a) elevates the stage to IIIA. [cite:Harrison 21e Ch 342], [cite:Robbins 10e Ch 24] 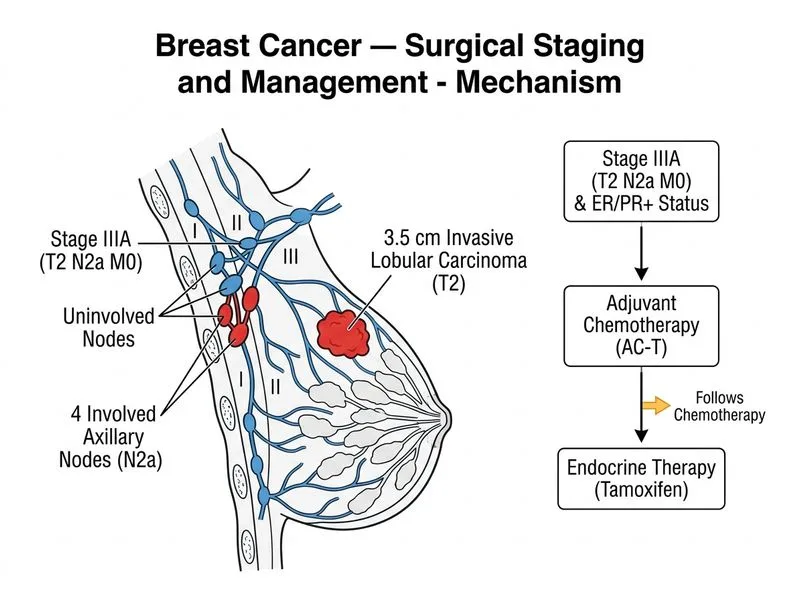
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.