## Knudson's Two-Hit Hypothesis in BRCA1-Associated Breast Cancer ### Clinical Context: Germline vs. Somatic Mutations **Key Point:** This patient carries a **germline BRCA1 mutation** (inherited, present in every cell). The loss of BRCA1 protein expression **specifically in tumor cells** (while normal tissue retains it) indicates a **second somatic event** — this is Knudson's two-hit model. ### The Two-Hit Hypothesis ```mermaid flowchart TD A[Inherited germline BRCA1 mutation<br/>nonsense mutation]:::outcome --> B[Heterozygous state<br/>one mutant, one wild-type allele]:::outcome B --> C{Somatic event in tumor cells}:::decision C -->|Loss of wild-type allele<br/>LOH| D[Homozygous mutant state<br/>Complete BRCA1 loss]:::action C -->|Epigenetic silencing<br/>methylation| E[Silenced wild-type allele<br/>Non-functional]:::action C -->|Gene conversion| F[Both alleles mutant]:::action D --> G[No functional BRCA1 protein<br/>Homologous recombination defect]:::urgent G --> H[Genomic instability<br/>Double-strand breaks unrepaired]:::urgent H --> I[Malignant transformation<br/>Aggressive phenotype]:::urgent ``` ### Why LOH (Loss of Heterozygosity) Is the Answer **High-Yield:** The immunohistochemistry findings are diagnostic: - **Normal tissue:** BRCA1 protein present (wild-type allele functional) - **Tumor cells:** BRCA1 protein absent (complete loss) This pattern is pathognomonic for **loss of the remaining wild-type allele** in the tumor. The mechanisms include: 1. **Chromosomal deletion** — loss of chromosome 17q (BRCA1 locus) 2. **Mitotic recombination** — homologous recombination between sister chromatids 3. **Gene conversion** — replacement of wild-type sequence with mutant sequence 4. **Nondisjunction** — loss of the chromosome carrying the wild-type allele ### Comparison of Mechanisms | Mechanism | Haploinsufficiency | LOH | Epigenetic Silencing | Dominant-Negative | |-----------|-------------------|-----|----------------------|-------------------| | **Protein level in tumor** | ~50% of normal | 0% (absent) | 0% (absent) | Abnormal complex | | **IHC pattern** | Reduced staining | Negative staining | Negative staining | Positive staining | | **Knudson two-hit?** | No (one hit) | Yes (two hits) | Yes (two hits) | No | | **Frequency in BRCA1 tumors** | Rare | ~70% | ~20% | Very rare | | **Functional consequence** | Partial DNA repair | Complete loss | Complete loss | Partial loss | **Clinical Pearl:** BRCA1 is a **recessive tumor suppressor** — one mutant allele is tolerated in normal cells (haploinsufficiency does not cause cancer), but loss of both alleles is required for malignant transformation. This is why germline BRCA1 carriers have high penetrance but do not develop cancer in all cells — only those that acquire a second somatic hit. ### Why BRCA1 Loss Drives Malignancy BRCA1 is essential for: 1. **Homologous recombination (HR)** — repair of double-strand breaks (DSBs) 2. **Cell cycle checkpoint control** — S/G2 checkpoint via p53 interaction 3. **Transcriptional regulation** — tumor suppression via p21 induction Loss of BRCA1 → Defective HR → Accumulation of DSBs → Genomic instability → Additional oncogenic mutations (TP53, RAS, PIK3CA) → Aggressive breast cancer **Mnemonic:** **BRCA-LOH** — BRCA1 Loss Of Heterozygosity is the classic two-hit mechanism in hereditary breast cancer. ### Why This Is Distinct from Haploinsufficiency **Warning:** Do not confuse haploinsufficiency with two-hit tumor suppression: - **Haploinsufficiency** = one mutant allele produces insufficient gene product (e.g., some dosage-sensitive genes like PTEN in Cowden syndrome) - **Recessive tumor suppression** = one mutant allele is silent; both alleles must be lost for cancer (e.g., BRCA1, RB, TP53) BRCA1 is a **recessive tumor suppressor**, not haploinsufficient — the wild-type allele produces enough protein to maintain DNA repair in normal cells. Only when both alleles are lost does malignancy develop. 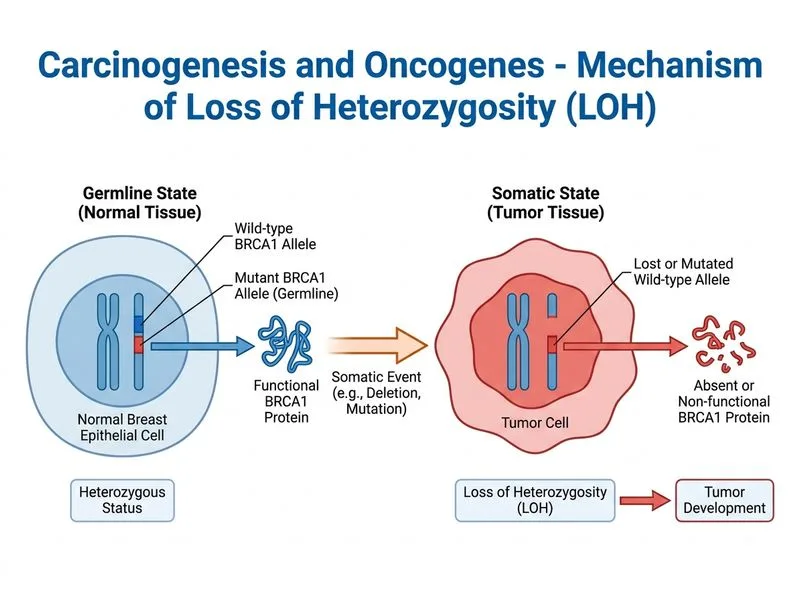
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.