## Management of Post-CCRT Morbidity in Cervical Cancer **Clinical Context:** This patient has completed definitive CCRT for stage IB2 cervical cancer with complete response. The presenting symptoms—vaginal stenosis and recurrent UTIs—are **late iatrogenic toxicities** of radiotherapy, not indicators of residual/recurrent disease (imaging is clear). **Correct Answer: Serial Vaginal Dilations + Topical Estrogen** **Key Point:** Vaginal stenosis is a common late effect of pelvic radiotherapy (incidence 5–25% in cervical cancer survivors). Management is **conservative and preventive**: - **Serial vaginal dilations** (3–4 times weekly) prevent adhesion formation and maintain vaginal patency - **Topical estrogen** (cream or tablets) restores mucosal health, improves lubrication, and reduces stenosis progression - **Routine surveillance imaging** (6–12 monthly) detects recurrence early - This approach preserves quality of life and sexual function without additional toxicity **High-Yield:** Vaginal stenosis management is **non-surgical** unless severe stricture or complete obliteration occurs. Early intervention prevents need for surgical reconstruction. **Clinical Pearl:** The combination of vaginal stenosis + recurrent UTIs suggests **radiation-induced changes** (fibrosis, reduced vascularity) rather than active disease. Estrogen deficiency (from ovarian damage) worsens mucosal atrophy. ## Why Each Distractor Is Wrong **Option 1 (Adjuvant Brachytherapy):** - Brachytherapy is part of **primary treatment** for cervical cancer, not an adjuvant modality after CCRT - Patient has already received full-dose external beam RT + brachytherapy is typically integrated into CCRT protocol - Adding brachytherapy post-CCRT would cause **severe cumulative toxicity** (bowel perforation, fistula, hemorrhage) without survival benefit when disease is in complete remission - **Contraindicated** in this clinical scenario **Option 2 (Systemic Chemotherapy with Bevacizumab):** - Bevacizumab is indicated for **recurrent or metastatic cervical cancer**, not for adjuvant therapy in stage IB2 with complete response - No survival benefit for adjuvant bevacizumab in early-stage disease post-CCRT - Unnecessary toxicity (hypertension, proteinuria, GI perforation risk) in a patient with no evidence of disease - NCCN/FIGO guidelines do not recommend adjuvant systemic therapy for stage IB2 with complete CCRT response **Option 3 (Total Abdominal Hysterectomy + BSO):** - Hysterectomy is **not indicated** after successful CCRT for cervical cancer - Increases morbidity (bowel adhesions, fistula risk) in a previously irradiated pelvis without improving survival - Ovarian preservation (in premenopausal women) is preferred to avoid surgical menopause and long-term HRT risks - Surgery is reserved for **persistent/recurrent disease** (exenteration) or as primary treatment in select early-stage cases (not post-CCRT) --- **Summary Table:** | Intervention | Indication | Status in This Case | |---|---|---| | Serial dilations + topical estrogen | Late radiation toxicity (stenosis) | ✓ **Appropriate** | | Adjuvant brachytherapy | Primary treatment integration | ✗ Already given; cumulative toxicity | | Bevacizumab | Recurrent/metastatic disease | ✗ No disease; no survival benefit | | Hysterectomy | Persistent/recurrent disease | ✗ Complete response; increases morbidity | 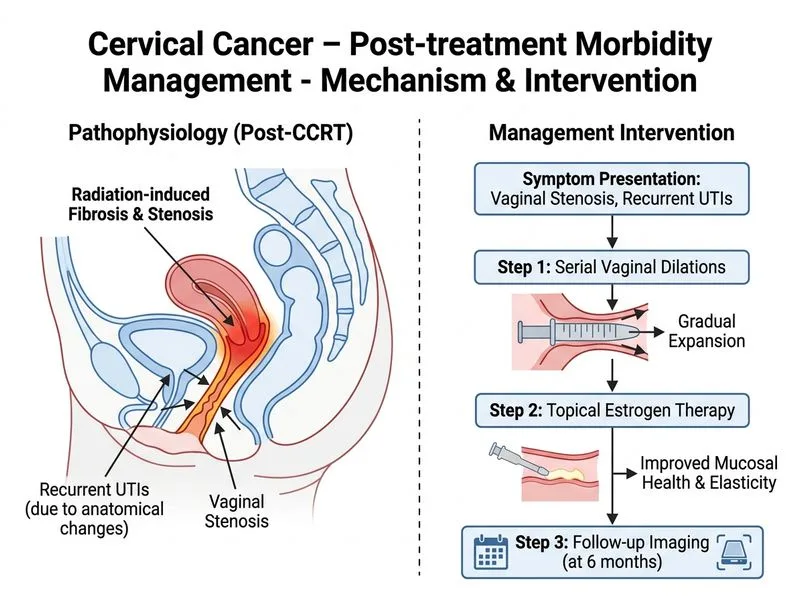
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.