## FIGO Staging and Treatment Algorithm ```mermaid flowchart TD A[Cervical Cancer Diagnosed]:::outcome --> B{Stage?}:::decision B -->|IA1-IB1| C[Radical hysterectomy or CCRT]:::action B -->|IB2-IIA| D[CCRT + Brachytherapy]:::action B -->|IIB-IVA| E[CCRT + Brachytherapy]:::action B -->|IVB| F[Chemotherapy + Palliative RT]:::action D --> G[Superior OS vs RT alone]:::outcome E --> H[Superior OS vs RT alone]:::outcome ``` ## Stage IIB Cervical Cancer: Treatment Rationale **Key Point:** Stage IIB cervical cancer (parametrial invasion without pelvic sidewall involvement) is a locally advanced disease that requires multimodal therapy. The standard of care is **concurrent chemoradiation (CCRT) followed by intracavitary brachytherapy**. **High-Yield:** The landmark GOG-120 and RTOG-90-01 trials (1999) demonstrated that concurrent cisplatin-based chemotherapy with external beam radiation therapy (EBRT) followed by brachytherapy improves overall survival (OS) by 30–50% compared to radiation alone in locally advanced cervical cancer (Stages IB2–IVA). ## Treatment Components | Component | Details | |-----------|----------| | **EBRT** | 45–50 Gy to pelvis over 5–6 weeks | | **Chemotherapy** | Cisplatin 40 mg/m² weekly during EBRT (5–6 cycles) | | **Brachytherapy** | Intracavitary HDR brachytherapy: 5–6 Gy × 5 fractions to point A after EBRT | | **Total EQD2** | ~80–90 Gy to point A (biologically equivalent dose) | ## Why NOT Surgery? **Clinical Pearl:** Radical hysterectomy with pelvic lymph node dissection is appropriate for **Stage IB1 disease only** (tumor ≤4 cm, no parametrial invasion). Stage IIB with parametrial invasion is surgically unresectable with adequate margins and has worse outcomes if surgery is attempted alone. Neoadjuvant chemotherapy followed by surgery is not standard and lacks robust evidence in this setting. ## Why NOT EBRT Alone? EBRT monotherapy has significantly lower cure rates (~50% 5-year OS) compared to CCRT + brachytherapy (~65–75% 5-year OS) in Stage IIB disease. Concurrent cisplatin acts as a radiosensitizer and provides systemic coverage for occult metastases. ## Citation [cite:GOG-120 and RTOG-90-01 Trials; FIGO Cervical Cancer Staging 2009; Obstetric and Gynaecological Society of India Cervical Cancer Management Guidelines] 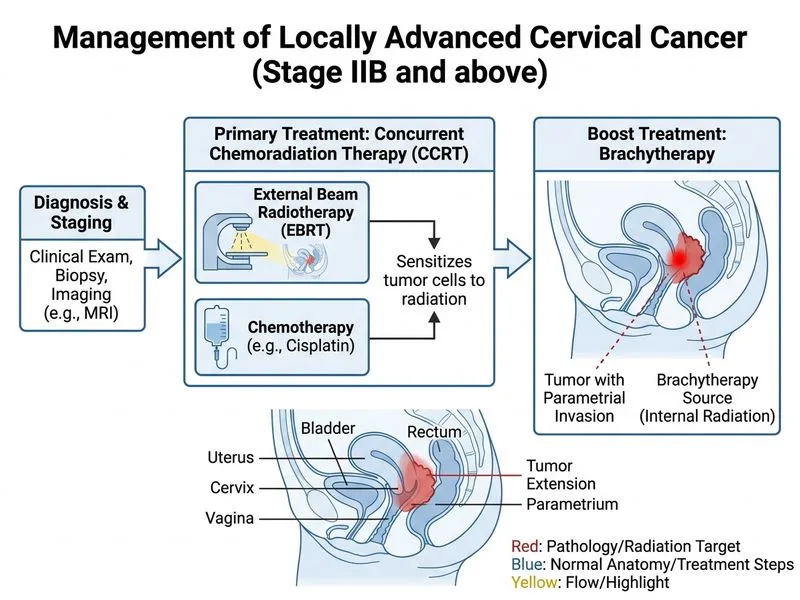
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.