## Classification of Chemical Injuries **Key Point:** Alkali burns cause more severe and progressive ocular damage compared to acid burns due to their mechanism of action. ### Why Alkalis Are More Destructive 1. **Penetration depth**: Alkalis saponify lipids in cell membranes, allowing deeper penetration into ocular tissues 2. **Progressive damage**: Alkali injuries continue to cause tissue damage even after the initial exposure (hours to days) 3. **Collagen dissolution**: Alkalis denature collagen, leading to corneal melting and perforation 4. **Anterior chamber involvement**: Alkalis readily penetrate through the cornea into the anterior chamber, affecting the iris, lens, and angle structures ### Comparison: Acid vs. Alkali Burns | Feature | Acid Burns | Alkali Burns | |---------|-----------|-------------| | **Mechanism** | Protein coagulation | Saponification of lipids | | **Penetration** | Superficial (coagulum acts as barrier) | Deep and progressive | | **Corneal opacity** | Usually reversible | Often irreversible | | **Anterior chamber** | Rarely involved | Frequently involved | | **Prognosis** | Better | Worse | | **Timeline** | Acute phase only | Acute + chronic phases | **High-Yield:** Sodium hydroxide (NaOH), potassium hydroxide (KOH), ammonia, and lime are common alkalis causing severe eye injuries. Lime (calcium oxide) is particularly dangerous because it also causes thermal injury. **Clinical Pearl:** In alkali burns, the initial appearance may be deceptively mild (clear cornea), but progressive opacification and corneal melting occur over subsequent hours due to ongoing collagen destruction. ## Management Principle Alkali injuries require aggressive and prolonged irrigation (at least 15–30 minutes) followed by chelation therapy with agents like EDTA to remove alkali cations from tissues. 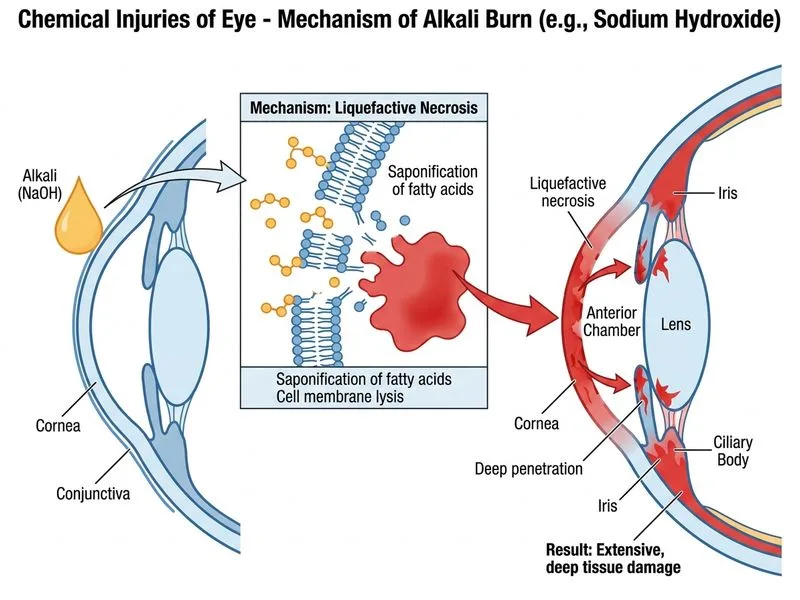
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.