## Cytogenetic Abnormalities in Childhood AML **Key Point:** t(15;17) PML-RARA is the hallmark cytogenetic abnormality of acute promyelocytic leukemia (APL, AML-M3) and confers excellent prognosis with ATRA and arsenic trioxide therapy. ### Recurrent Cytogenetic Abnormalities in Childhood AML | Abnormality | AML Subtype | Prognosis | Treatment Response | |---|---|---|---| | t(15;17) PML-RARA | APL (M3) | Excellent | ATRA + arsenic trioxide; >90% cure | | t(8;21) RUNX1-RUNX1T1 | AML-M2 (AML with maturation) | Favorable | Intensive chemotherapy; 60–70% EFS | | inv(16) CBFB-MYH11 | AML-M4Eo (AML with eosinophilia) | Favorable | Intensive chemotherapy; 60–70% EFS | | Complex karyotype | Various | Adverse | Poor chemotherapy response; <30% EFS | | Monosomy 7 | Various | Adverse | High-risk disease; requires allogeneic HSCT | **High-Yield:** APL is a medical emergency due to risk of disseminated intravascular coagulation (DIC) and life-threatening hemorrhage, but it is also the most curable AML subtype when treated with ATRA + arsenic trioxide. **Clinical Pearl:** The PML-RARA fusion protein acts as a ligand-dependent transcriptional repressor. ATRA binds to the fusion protein and causes differentiation of leukemic promyelocytes into mature granulocytes, leading to remission. **Mnemonic:** **APL = ATRA Promyelocytic Leukemia** — remember that APL is the one AML that responds to ATRA, not chemotherapy alone. ### Mechanism of ATRA in APL 1. ATRA binds to the PML-RARA fusion protein. 2. Causes degradation of the fusion protein and restoration of normal retinoic acid signaling. 3. Leukemic promyelocytes differentiate into mature, non-proliferating granulocytes. 4. Combination with arsenic trioxide (which targets PML protein) achieves cure in >90% of cases. 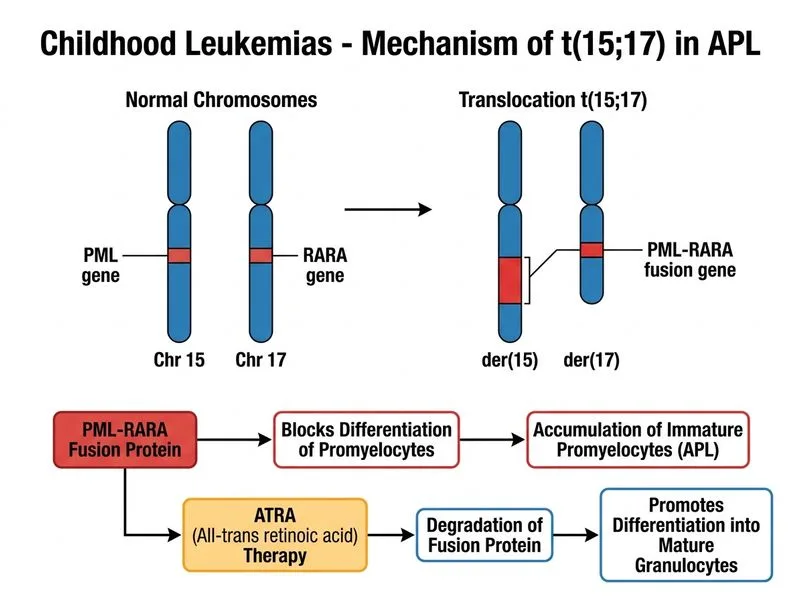
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.