## Management of Acute Promyelocytic Leukemia (APL): Targeted Therapy ### Diagnosis: APL (AML-M3) **Key Point:** The t(15;17) translocation is pathognomonic for acute promyelocytic leukemia (APL), a subtype of AML characterized by abnormal promyelocytes and a unique treatment paradigm. **High-Yield:** APL is the ONLY subtype of AML where targeted differentiation therapy (ATRA + arsenic trioxide) has replaced conventional chemotherapy as first-line induction. ### Molecular Basis of APL The t(15;17) translocation fuses the **PML gene** (chromosome 17) with the **RARA gene** (retinoic acid receptor-α, chromosome 15), creating the PML-RARA fusion protein. This fusion: - Blocks normal myeloid differentiation - Represses genes required for maturation - Is exquisitely sensitive to ATRA and arsenic trioxide ### Standard Induction Regimen for APL | Agent | Mechanism | Dosing | Role | |---|---|---|---| | ATRA (all-trans retinoic acid) | Binds PML-RARA; promotes differentiation | 45 mg/m²/day in 2 doses | Induces maturation | | Arsenic trioxide (ATO) | Causes PML-RARA degradation and apoptosis | 0.15 mg/kg/day IV | Synergistic with ATRA | | Supportive care | Transfusions, DIC prophylaxis, electrolyte monitoring | — | Prevents hemorrhage and metabolic complications | **Clinical Pearl:** APL is highly curable with ATRA + ATO, with complete remission rates >90% and 5-year overall survival >80% in children. This is a dramatic contrast to other AML subtypes. ### Critical Supportive Measures **Warning:** APL has two life-threatening complications that must be managed aggressively: 1. **Disseminated Intravascular Coagulation (DIC)** - Present at diagnosis in ~80% of cases - Caused by release of procoagulant from abnormal promyelocytes - Management: Fresh frozen plasma, cryoprecipitate, low-dose heparin if severe 2. **Differentiation Syndrome (formerly ATRA syndrome)** - Occurs during ATRA/ATO therapy as blasts differentiate - Features: fever, respiratory distress, pulmonary infiltrates, hypoxia, weight gain - Management: Dexamethasone 10 mg IV Q12H; may need to hold ATRA temporarily ### Why Not the Other Options? **Option 0 (Standard AML chemotherapy):** Daunorubicin + cytarabine is the backbone for non-APL AML. In APL, ATRA + ATO is superior because it targets the underlying molecular defect (PML-RARA) and avoids the cardiotoxicity and myelosuppression of conventional chemotherapy. **Option 2 (Immediate allogeneic SCT):** Stem cell transplantation is not part of first-line induction for APL. It is reserved for relapsed or refractory disease or for high-risk patients (e.g., high WBC count >10,000/μL at diagnosis). Most APL patients achieve durable remission with ATRA + ATO alone. **Option 3 (High-dose cytarabine + mitoxantrone):** This is salvage therapy for relapsed/refractory AML, not first-line for APL. ### Treatment Algorithm for APL ```mermaid flowchart TD A["Newly Diagnosed APL<br/>(t15;17)"]:::outcome --> B["Assess DIC status<br/>Transfusion support"]:::action B --> C["Start ATRA + ATO<br/>+ Dexamethasone cover"]:::action C --> D{"Differentiation<br/>syndrome?"}:::decision D -->|Yes| E["High-dose dexamethasone<br/>Hold ATRA if severe"]:::action D -->|No| F["Continue ATRA + ATO<br/>Monitor CBC, coagulation"]:::action E --> F F --> G{"Complete remission<br/>achieved?"}:::decision G -->|Yes| H["Consolidation + maintenance<br/>ATRA + ATO"]:::action G -->|No| I["Salvage therapy<br/>Consider SCT"]:::urgent H --> J["Long-term follow-up<br/>Cure rate >80%"]:::outcome ``` [cite:Gupta & Sarin Pediatric Hematology-Oncology Ch 6; NCCN AML Guidelines 2023; Sanz et al. Leukemia 2019] 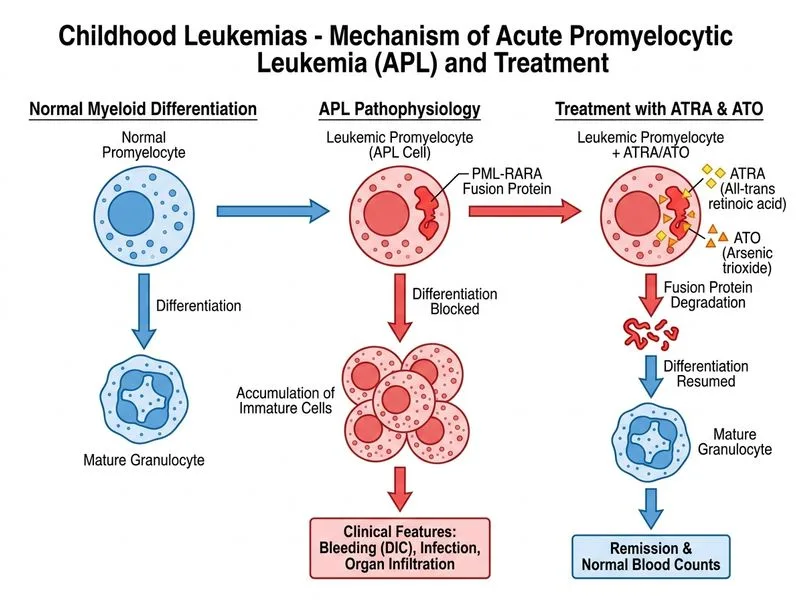
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.