## Acute Promyelocytic Leukemia (APL) and t(15;17) **Key Point:** The t(15;17) translocation resulting in PML-RARA fusion is pathognomonic for acute promyelocytic leukemia (APL, AML-M3). This abnormality is highly sensitive to all-trans retinoic acid (ATRA) and arsenic trioxide (ATO), which induce differentiation and apoptosis of leukemic blasts. ### Molecular Basis and Treatment Response **High-Yield:** The PML-RARA fusion protein acts as a dominant-negative retinoic acid receptor, blocking myeloid differentiation. ATRA and ATO disrupt this fusion protein, restoring differentiation and triggering apoptosis. **Mnemonic:** **APL = t(15;17) = ATRA/ATO** — remember that APL is the only AML subtype with a specific targeted therapy that dramatically improves survival. ### APL: Cytogenetics and Prognosis | Feature | Details | |---|---| | **Cytogenetics** | t(15;17) PML-RARA (>95% of APL cases) | | **Morphology** | Abnormal promyelocytes with Auer rods (often multiple, "faggot cells") | | **Coagulopathy** | DIC (disseminated intravascular coagulation) common; life-threatening hemorrhage | | **Treatment** | ATRA + ATO ± chemotherapy | | **Cure Rate** | 80–90% with modern ATRA/ATO-based regimens | | **Relapse Risk** | Low if PML-RARA PCR negativity achieved | **Clinical Pearl:** APL is a medical emergency due to severe coagulopathy (DIC) and high risk of intracranial hemorrhage. Early recognition and immediate initiation of ATRA (even before cytogenetic confirmation) is life-saving. ### Why Other Abnormalities Are Not APL - **t(8;21) and inv(16):** These are core binding factor (CBF) AMLs—favorable prognosis but NOT APL. They respond to chemotherapy, not ATRA/ATO. - **FLT3-ITD:** A mutation conferring poor prognosis in AML; not specific to APL and not targeted by ATRA/ATO. 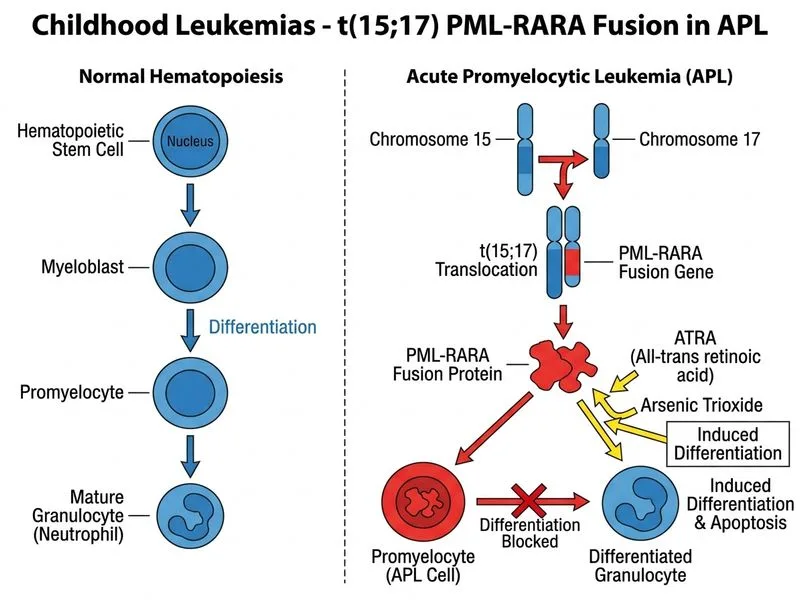
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.