## Clinical Context This patient has CKD stage 4 with evidence of secondary hyperparathyroidism (sHPT) and mineral-bone disorder (MBD): - **Elevated PTH** (285 pg/mL, normal 15–65) indicates sHPT - **Hyperphosphatemia** (5.8 mg/dL, normal 2.5–4.5) — phosphate retention begins at eGFR <60 - **Hypocalcemia** (8.1 mg/dL, normal 8.5–10.5) — reflects reduced calcitriol production and hyperphosphatemia - **Anemia** (Hb 9.2 g/dL) — multifactorial (erythropoietin deficiency, uremia, iron deficiency) ## High-Yield: CKD-MBD Management Algorithm ```mermaid flowchart TD A[CKD Stage 4, eGFR 18]:::outcome --> B{Biochemistry abnormal?}:::decision B -->|Yes: High PTH, High PO4, Low Ca| C[Initiate MBD therapy]:::action C --> D[Phosphate binder first-line]:::action C --> E[Calcitriol if Ca low + PTH elevated]:::action A --> F{Hemoglobin <10?}:::decision F -->|Yes| G[Start ESA + iron]:::action A --> H{eGFR approaching 15-20?}:::decision H -->|Yes| I[Refer for vascular access planning]:::action I --> J[Fistula creation 3-6 months before dialysis]:::action D --> K[Recheck PO4, Ca, PTH in 4-6 weeks]:::action ``` ## Key Point: Phosphate Binder Selection | Agent | Mechanism | Advantage | Disadvantage | |-------|-----------|-----------|---------------| | **Calcium-based** (CaCO₃, CaAc) | Binds PO₄ in gut | Cheap, raises Ca | Risk of hypercalcemia, vascular calcification | | **Non-calcium** (Sevelamer, Lanthanum) | Binds PO₄ in gut | No hypercalcemia risk | Expensive, GI side effects | | **Iron-based** (Sucroferric oxyhydroxide) | Binds PO₄ in gut | No Ca/hypercalcemia | Newer, less data | **High-Yield:** In CKD stage 4 with hypocalcemia and elevated PTH, **calcium-based binders are acceptable** as first-line (e.g., calcium carbonate 1–2 g TDS with meals) because the patient needs calcium supplementation. Non-calcium binders are reserved for hypercalcemic patients. ## Calcitriol Indications **Key Point:** Calcitriol (active vitamin D, 1,25-dihydroxyvitamin D₃) is indicated when: - PTH is elevated (>65 pg/mL) **AND** - Serum calcium is low or low-normal (<8.5 mg/dL) **AND** - eGFR is <30 mL/min/1.73m² This patient meets all criteria. Dose: typically 0.25–0.5 μg BD or TDS; monitor Ca and PTH every 4–6 weeks. ## Anemia Management **Clinical Pearl:** ESA (erythropoiesis-stimulating agent) initiation is indicated when: - Hemoglobin <10 g/dL (this patient: 9.2 g/dL) ✓ - Symptoms of anemia or CKD progression - After iron stores are assessed (ferritin, TSAT) Target hemoglobin: 10–11.5 g/dL (avoid overcorrection >12 g/dL due to cardiovascular risk). ## Vascular Access Planning **High-Yield:** **Vascular access creation should begin when eGFR approaches 15–20 mL/min/1.73m²** (this patient is at eGFR 18, so timing is appropriate). Fistula maturation takes 3–6 months; early referral prevents urgent-start dialysis and reduces infection risk. ## Integrated Next Steps 1. **Start phosphate binder** (calcium carbonate 1–2 g TDS with meals) 2. **Initiate calcitriol** (0.25 μg BD; titrate based on PTH and Ca) 3. **Start ESA** (e.g., epoetin alfa 50 IU/kg SC 2–3×/week or darbepoetin alfa 0.45 μg/kg SC weekly) + iron supplementation 4. **Refer to nephrologist for vascular access planning** (arteriovenous fistula preferred) 5. **Recheck biochemistry in 4 weeks** (PTH, Ca, PO₄, Hb) 6. **Assess dialysis readiness** (education, modality choice, psychosocial support) ## Why Option 2 Is Correct Option 2 is the most comprehensive and guideline-aligned approach because it addresses all three major complications of CKD stage 4: - **MBD:** phosphate binder + calcitriol - **Anemia:** ESA + iron - **Preparation for RRT:** vascular access referral [cite:KDIGO 2017 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease; Harrison 21e Ch 279] 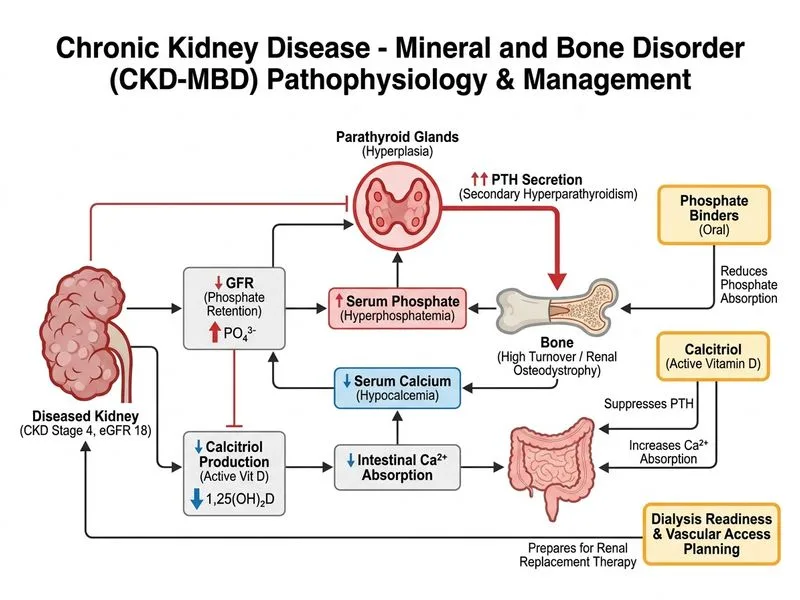
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.