## Management of Newly Diagnosed CML in Chronic Phase **Key Point:** Imatinib mesylate (a BCR-ABL tyrosine kinase inhibitor) is the standard first-line therapy for newly diagnosed CML in chronic phase, regardless of age or comorbidities, provided there are no contraindications. ### Rationale for Imatinib as First-Line Imatinib has revolutionized CML management since its approval. It targets the constitutively active BCR-ABL fusion protein, inducing apoptosis and inhibiting proliferation of leukemic cells. **High-Yield:** The standard starting dose is 400 mg once daily for chronic phase CML. This achieves complete cytogenetic response (CCyR) in 80–90% of patients and major molecular response (MMR) in 60–70% within 12 months. ### Treatment Milestones & Monitoring | Timepoint | Target Response | Investigation | |-----------|-----------------|----------------| | 3 months | BCR-ABL transcript reduction to <10% | | Quantitative RT-PCR (qRT-PCR) | | 6 months | Major molecular response (MMR, <0.1%) | qRT-PCR | | 12 months | Complete molecular response (CMR, undetectable) | qRT-PCR | **Clinical Pearl:** Response assessment is done by quantitative RT-PCR (qRT-PCR) of BCR-ABL transcript levels, not by cytogenetics alone. Failure to achieve MMR by 12 months warrants investigation for resistance mutations (especially T315I) and consideration of second-generation TKIs (dasatinib, nilotinib, ponatinib). ### Pre-Treatment Considerations 1. Baseline cardiac assessment (imatinib can rarely cause cardiomyopathy) 2. Renal and hepatic function tests 3. Tumor lysis syndrome (TLS) prophylaxis: allopurinol or febuxostat + hydration (especially if WBC > 100,000/μL or high blast percentage) 4. Baseline BCR-ABL quantitation for response monitoring **Warning:** Do NOT initiate hydroxyurea as monotherapy in the modern era — it is inferior to TKI therapy and delays optimal disease control. Hydroxyurea is reserved for TKI-intolerant patients or as a bridge therapy in rare circumstances. ## Why Other Options Are Incorrect **Splenectomy** is not routinely indicated in CML. While splenomegaly is common, splenectomy does not alter the natural history of the disease and is reserved for symptomatic massive splenomegaly (pain, early satiety) refractory to medical therapy or for rare complications like splenic infarction. **Allogeneic HSCT** is no longer the first-line treatment for chronic phase CML. It is reserved for: - TKI-resistant disease (BCR-ABL mutations, primary or secondary resistance) - Accelerated phase or blast crisis - Patients with poor prognostic factors (Sokal or Hasford score) who fail to achieve MMR by 12 months - Young patients with matched sibling donors and high-risk disease (discussed on case-by-case basis) The shift from HSCT to TKI-first strategy has dramatically improved outcomes and quality of life, with median overall survival now exceeding 20+ years in chronic phase CML. [cite:Harrison 21e Ch 104] 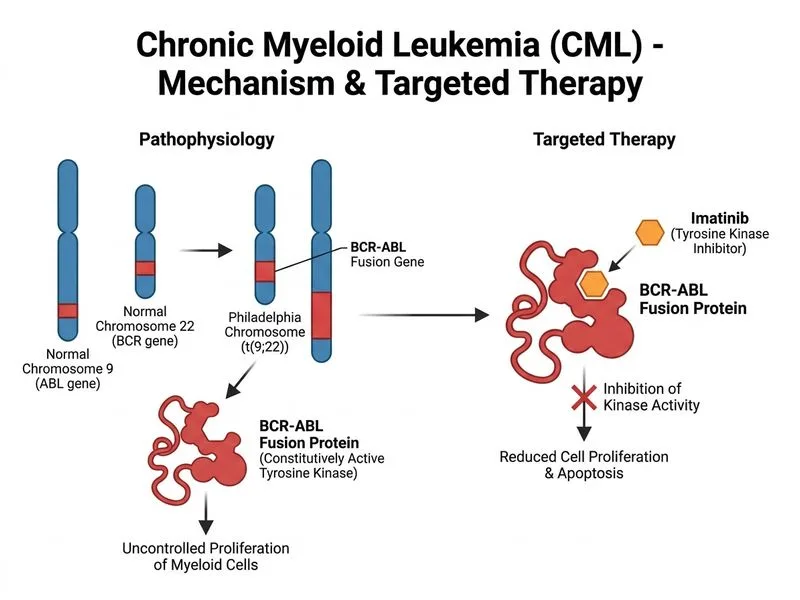
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.