## Clinical Context The presentation of a 6-year-old with cerebellar signs (ataxia, truncal instability), raised intracranial pressure (papilledema, morning vomiting), and a midline posterior fossa mass is classic for **medulloblastoma** or **ependymoma**. The **hypointense T2 signal in the center** with perilesional edema strongly suggests **hemorrhage or calcification** within the lesion — features that require susceptibility-sensitive sequences for optimal characterization. ## Why SWI and Gradient Echo Are Optimal **Key Point:** The stem specifically asks about characterizing the **internal composition** of the lesion (the hypointense T2 center) and guiding surgical planning based on that characterization. SWI (Susceptibility-Weighted Imaging) and gradient echo sequences are the most sensitive MRI techniques for detecting **paramagnetic substances**, including: - **Hemosiderin / blood products** (from prior hemorrhage — "blooming artifact") - **Calcification** (hypointense on T2, further confirmed on SWI) - **Iron deposition** and deoxygenated blood - **Microhemorrhages** not visible on conventional T2/FLAIR **High-Yield (per Osborn's Brain Imaging):** Medulloblastomas frequently demonstrate hemorrhage and calcification. SWI is superior to conventional T2* for detecting microhemorrhages and calcification, directly informing the surgeon about intraoperative bleeding risk and areas of necrosis/calcification that affect resection strategy. **Clinical Pearl:** In pediatric posterior fossa tumors, the hypointense T2 center is the key discriminating finding in this stem. SWI/gradient echo directly addresses this finding. DTI/tractography, while valuable for white matter preservation, does NOT characterize the internal lesion composition — it maps surrounding tracts. The question's primary intent is lesion characterization, with surgical planning as a downstream benefit of that characterization. ## Surgical Planning Advantage of SWI - Identifies hemorrhagic foci → predicts intraoperative bleeding risk - Delineates calcified vs. necrotic core → guides debulking approach - Detects leptomeningeal microhemorrhagic deposits (staging relevance in medulloblastoma) ## Why Other Options Are Incorrect | Modality | Limitation | |---|---| | **DTI + Fiber Tractography** | Maps white matter tracts adjacent to tumor (critical for eloquent area preservation), but does NOT characterize the internal composition (hemorrhage/calcification) of the lesion itself — does not address the hypointense T2 center | | **MR Spectroscopy + PWI** | Assesses metabolic profile (elevated choline, reduced NAA, choline:creatinine ratio) and tumor perfusion/rCBV for grading; valuable for tumor typing but does NOT specifically characterize hemorrhage or calcification — secondary to SWI for this specific imaging finding | | **Contrast-enhanced CT** | Detects calcification well, but is **inferior to SWI for hemorrhage detection**; adds ionizing radiation in a pediatric patient; MRI already provides superior soft tissue contrast; CT is not the "further characterization" step when MRI workup is ongoing | **High-Yield:** Per Osborn's Brain Imaging and ACR guidelines, SWI is the sequence of choice when hemorrhage or calcification is suspected within a CNS tumor on conventional MRI — it is the logical next step when a hypointense T2 center is identified in a posterior fossa pediatric mass. **Mnemonic:** **SWI** = **S**usceptibility detects **W**hat's **I**nside — hemorrhage, iron, calcification. 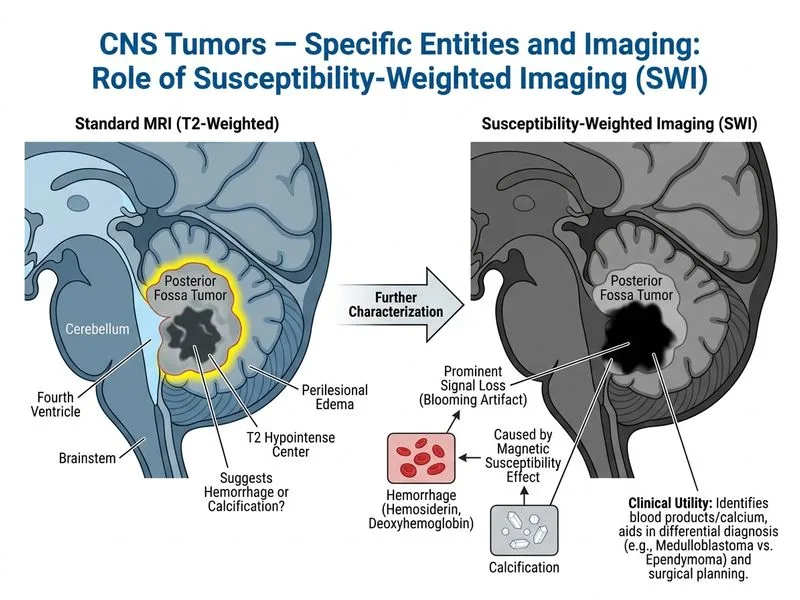
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.