## Ventricular Septal Defect (VSD) — Timing of Surgical Repair ### Clinical Presentation & Pathophysiology This infant presents with a **large, hemodynamically significant VSD**: - **Palpable thrill** — indicates significant turbulent flow across the defect - **Systolic ejection murmur** — blood flowing from left to right ventricle during ventricular systole - **Widely split S~2~** — increased right ventricular stroke volume delays pulmonary valve closure - **Pulmonary congestion & cardiomegaly** — left-to-right shunt causes volume overload of both ventricles and pulmonary circulation - **Poor feeding & failure to thrive** — signs of congestive heart failure (CHF) in infancy - **Elevated pulmonary artery pressures** — chronic volume overload causes reactive pulmonary hypertension **Key Point:** This is a large VSD with hemodynamic significance. The question is not *whether* to repair, but *when* and *how*. ### Natural History of VSDs | VSD Size | Hemodynamic Significance | Natural Closure Rate | Management | |---|---|---|---| | **Small (restrictive)** | Minimal | 80–90% by age 5 years | Conservative; serial echo | | **Moderate** | Mild–moderate | 30–50% by age 5 years | Medical management; elective repair | | **Large (non-restrictive)** | Severe; CHF | <10% spontaneous closure | Medical management + planned surgical repair | **High-Yield:** Large VSDs with CHF symptoms require **medical stabilization followed by surgical repair**; they do NOT close spontaneously and carry high risk of Eisenmenger syndrome if left unrepaired. ### Medical Management Phase (Preoperative Stabilization) Before surgical repair, the infant is managed medically to: 1. **Reduce pulmonary edema & improve feeding:** Diuretics (furosemide) 2. **Reduce afterload & improve cardiac output:** ACE inhibitors (enalapril, lisinopril) 3. **Optimize contractility:** Digoxin (inotropic support) 4. **Nutritional support:** High-calorie feeds (120–150 kcal/kg/day) to promote growth **Clinical Pearl:** The goal of medical management is to allow the infant to grow to a suitable size and weight for surgery (typically 3–4 kg or 3–4 months of age), while preventing complications like severe pulmonary hypertension or recurrent respiratory infections. ### Timing of Surgical Repair ```mermaid flowchart TD A[Large VSD with CHF symptoms]:::outcome --> B[Start medical management]:::action B --> C[Diuretics + ACE inhibitors + Digoxin]:::action C --> D[Monitor growth & clinical status]:::action D --> E{Adequate growth<br/>3-4 kg or 3-4 months?}:::decision E -->|Yes| F[Elective surgical repair]:::action E -->|No| G[Continue medical therapy<br/>Reassess in 2-4 weeks]:::action G --> D D --> H{Deterioration:<br/>Severe PHT, recurrent<br/>infections, failure to thrive?}:::decision H -->|Yes| I[Urgent surgical repair<br/>or PA banding]:::urgent H -->|No| F F --> J[VSD closure<br/>Normal life expectancy]:::outcome ``` ### Why NOT Immediate Surgery? - **Risk:** Neonates and very young infants have higher perioperative mortality - **Benefit of waiting:** Allows infant to grow, develop, and tolerate surgery better - **Standard practice:** Elective repair at 3–4 months of age (or 3–4 kg body weight) is the benchmark ### Why NOT Pulmonary Artery Banding? - **PA banding** is reserved for: - **Multiple VSDs** (too numerous to close individually) - **Complex anatomy** (e.g., VSD with other major defects) - **Extreme prematurity or medical instability** (temporary measure) - In a straightforward large VSD, definitive closure is preferred over a staged approach ### Why NOT Watchful Waiting? - **Large VSDs do NOT close spontaneously** (<10% closure rate) - **Risk of Eisenmenger syndrome:** Chronic left-to-right shunt → reactive pulmonary hypertension → reversal of shunt → cyanosis and irreversible pulmonary vascular disease - **Symptoms warrant intervention:** Poor feeding, failure to thrive, and CHF indicate hemodynamic significance **Warning:** Confusing large VSDs (which require surgery) with small restrictive VSDs (which often close spontaneously) is a common exam trap. The presence of a thrill, wide split S~2~, and pulmonary congestion indicates a large, hemodynamically significant defect requiring repair. ## Why the Correct Answer Is Correct The standard management of a large, hemodynamically significant VSD in a 2-month-old is medical stabilization (diuretics, ACE inhibitors, digoxin) followed by elective surgical repair when the child reaches 3–4 kg body weight or 3–4 months of age. This approach balances the need for definitive closure (to prevent Eisenmenger syndrome) with the infant's need to grow and develop before undergoing surgery. 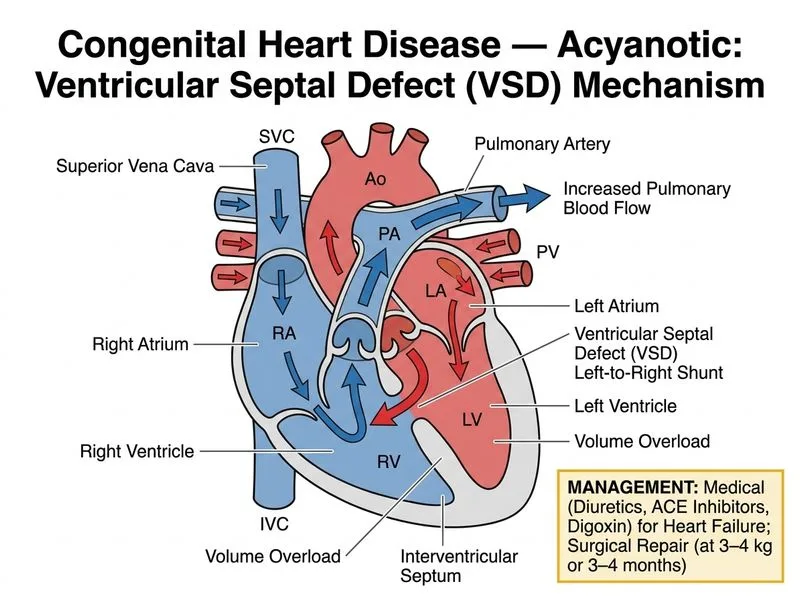
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.