## Ionic vs Non-Ionic Contrast Media: Key Discriminators ### Osmolality and Reaction Mechanisms **Key Point:** Ionic HOCM (osmolality 1400–1900 mOsm/kg) causes reactions primarily through **direct mast cell degranulation and chemotoxicity**, whereas non-ionic LOCM (osmolality 600–850 mOsm/kg) triggers reactions via **indirect mechanisms** like complement activation and bradykinin release. ### Comparison Table | Feature | Ionic HOCM | Non-Ionic LOCM | | --- | --- | --- | | **Osmolality** | 1400–1900 mOsm/kg | 600–850 mOsm/kg | | **Ionization** | Dissociates into cations + anions | Remains undissociated | | **Reaction Mechanism** | Direct mast cell degranulation; osmotic toxicity | Bradykinin, complement activation; chemotoxicity | | **Incidence of ADR** | 5–8% (overall); 0.2–0.4% severe | 0.4–0.7% (overall); 0.004% severe | | **Renal Risk** | Higher (osmotic diuresis, tubular damage) | Lower | | **Cardiac Arrhythmias** | Common (hyperkalemia from dissociation) | Rare | ### Why Osmolality Matters **High-Yield:** The **osmotic gradient** in HOCM (>1400 mOsm/kg) causes: 1. Water shift out of cells → cellular dehydration 2. Intravascular volume expansion → hypervolemia, pulmonary edema 3. Direct endothelial and mast cell injury Non-ionic LOCM avoids this osmotic stress but still triggers **chemotoxic reactions** through complement and contact system activation — a different pathophysiology. ### Clinical Pearl **Warning:** Do not confuse "low osmolality" with "no reaction risk." Non-ionic LOCM still causes 0.4–0.7% adverse reactions; they are simply **less severe and less frequent** than HOCM. The mechanism is chemotoxicity, not osmotic injury. ### Mnemonic **IONIC = Osmotic Injury**: **I**onic HOCM causes **O**smotic damage (mast cell degranulation, fluid shifts); **N**on-ionic LOCM causes **I**ndirect chemotoxic reactions (bradykinin, complement). 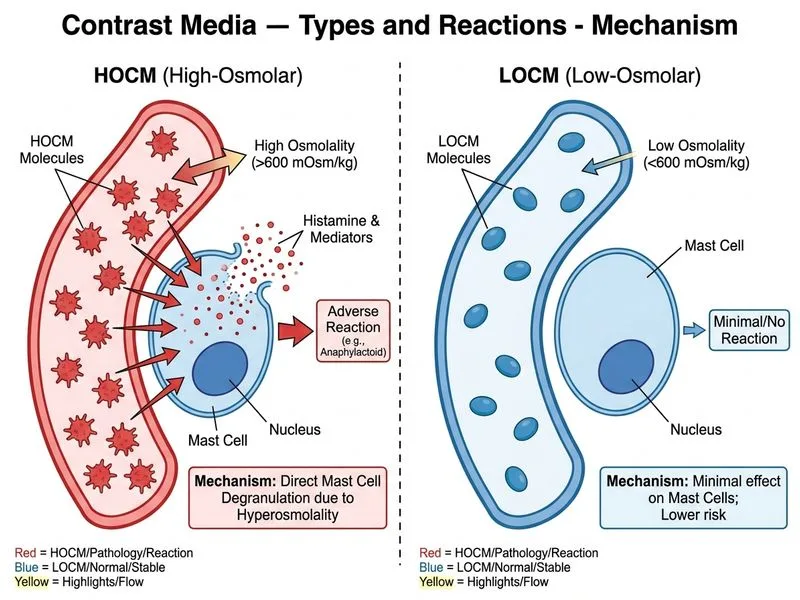
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.