## Classification of Radiographic Contrast Media **Key Point:** Contrast media are classified primarily by osmolality, which directly correlates with adverse reaction risk and hemodynamic effects. ### Osmolality-Based Classification | Classification | Osmolality (mOsm/kg) | Examples | Adverse Reaction Risk | |---|---|---|---| | **High-osmolality (HOCM)** | 1500–2400 | Diatrizoate, Meglumine | High (5–8%) | | **Low-osmolality (LOCM)** | 600–900 | Iopamidol, Iohexol, Ioversol | Low (0.2–0.7%) | | **Iso-osmolality (IOCM)** | ~290 | Iodixanol | Very low (<0.1%) | **High-Yield:** Diatrizoate is the classic HOCM agent — it is a **monomer** with a single iodine-carrying benzene ring, resulting in high osmolality relative to iodine content. The high osmolality causes: - Fluid shift into intravascular space - Transient hypervolemia and hypertension - Increased risk of contrast-induced nephropathy (CIN) - Higher incidence of allergic-like reactions ### Why HOCM is Rarely Used Today **Clinical Pearl:** HOCM agents are now largely obsolete in developed countries due to superior safety profile of LOCM and IOCM agents. They are occasionally retained in resource-limited settings or for specific applications (e.g., some interventional procedures where cost is prohibitive). **Mnemonic:** **HOCM = High Osmolality = High Harm** — remember that osmolality drives adverse effects more than iodine concentration alone. ### Mechanism of Osmolality-Related Toxicity High osmolality causes: 1. Osmotic diuresis and dehydration 2. Endothelial cell injury and increased vascular permeability 3. Increased blood viscosity 4. Renal tubular toxicity (especially in pre-existing renal disease) **Warning:** Do not confuse osmolality with iodine concentration — a LOCM agent can have high iodine content (e.g., iopamidol 370 mg I/mL) but low osmolality because it uses **dimeric** or **non-ionic** structures that reduce particle number. 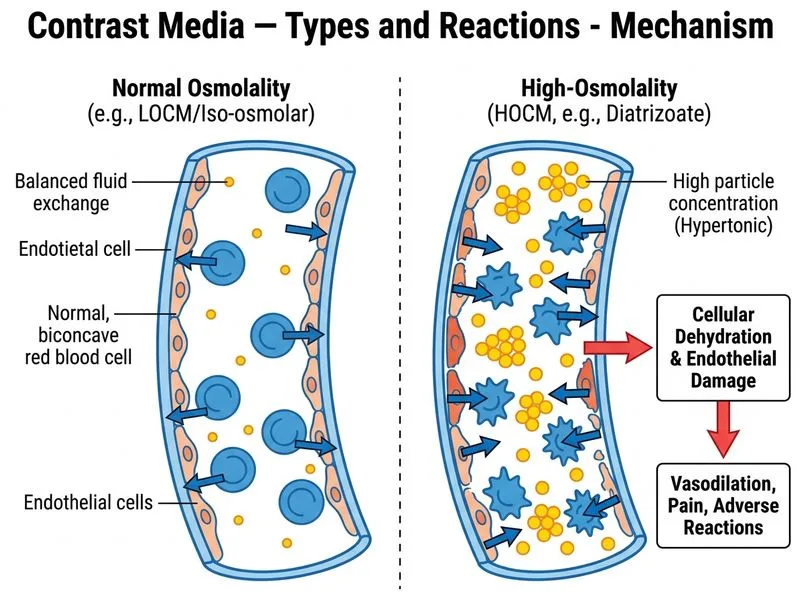
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.