## Distinguishing Feature: Osmolality and Reaction Incidence ### Osmolality as the Key Discriminator **Key Point:** Osmolality is the primary structural difference that predicts adverse reaction risk. Ionic HOCM has osmolality of 1400–2400 mOsm/kg (5–8 times plasma), whereas non-ionic LOCM has osmolality of 600–850 mOsm/kg (2–3 times plasma). ### Adverse Reaction Profile | Feature | Ionic HOCM | Non-ionic LOCM | |---------|-----------|----------------| | **Osmolality** | > 1400 mOsm/kg | < 800 mOsm/kg | | **Anaphylactoid reactions** | 1 in 5,000–14,000 | 1 in 40,000–200,000 | | **Chemotoxicity** | High (osmotic stress) | Low | | **Vasodilation** | Marked (hypotension) | Minimal | | **Cardiac arrhythmias** | Common | Rare | **High-Yield:** The osmotic gradient created by HOCM causes fluid shift into the intravascular space, endothelial damage, and mast cell degranulation — leading to a 10–20 fold higher rate of anaphylactoid reactions compared to LOCM. ### Clinical Pearl **Clinical Pearl:** Although both contain iodine, the osmolality difference — not iodine content — is the primary driver of adverse reactions. This is why non-ionic LOCM (e.g. iopamidol, iohexol) is now the standard for intravascular use, especially in high-risk patients (renal impairment, diabetes, elderly). **Mnemonic:** **HOCM = High Osmolality = High Harm** (anaphylactoid reactions, arrhythmias, renal toxicity). LOCM = Low osmolality = Lower risk. [cite:Harrison 21e Ch 474] 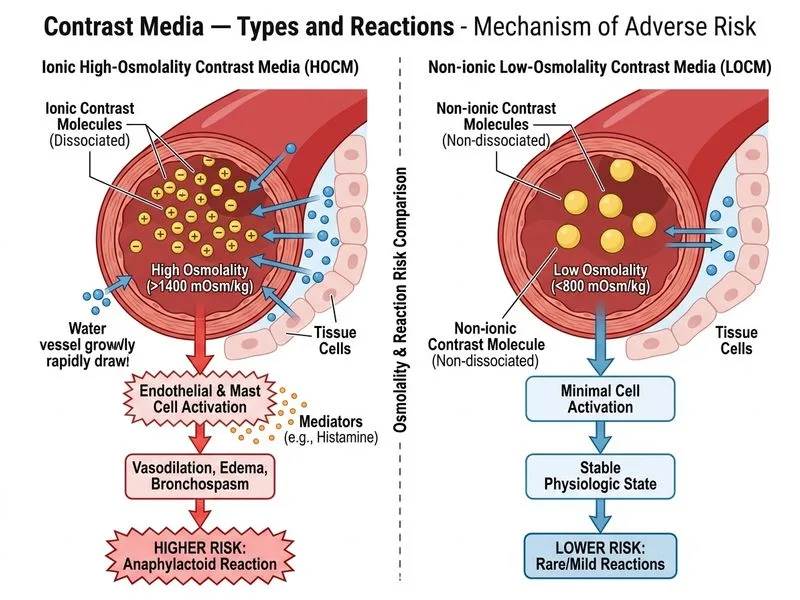
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.