## DIC-Induced Multi-Organ Failure in Metastatic Cancer ### Clinical Recognition of DIC with Organ Involvement **Key Point:** This patient has **overt DIC with acute respiratory distress syndrome (ARDS)** secondary to metastatic pancreatic cancer. The primary pathophysiology is **microthrombi formation in the pulmonary circulation**, not mechanical obstruction or cardiac failure. ### Laboratory Evidence of DIC | Parameter | Finding | Interpretation | |-----------|---------|----------------| | Platelets | 32,000/µL | Consumption | | PT/aPTT | 22 s / 52 s | Factor consumption | | Fibrinogen | 110 mg/dL | Consumption + fibrinolysis | | D-dimer | 12 µg/mL | Fibrin formation & degradation | | Schistocytes | Present | Mechanical hemolysis from fibrin strands | | PaO₂ | 58 mmHg | Pulmonary dysfunction | **High-Yield:** Pancreatic cancer is one of the **top 3 solid tumors** causing DIC (along with lung and gastric cancers). Cancer cells express tissue factor (TF) on their surface, directly activating the extrinsic coagulation pathway. ### Mechanism of Pulmonary Involvement in DIC ```mermaid flowchart TD A[Pancreatic cancer cells]:::outcome --> B[Express Tissue Factor]:::action B --> C[Extrinsic pathway activation]:::action C --> D[Thrombin generation]:::outcome D --> E[Fibrin deposition in lungs]:::urgent E --> F[Microthrombi in pulmonary capillaries]:::urgent F --> G[Ventilation-Perfusion mismatch]:::action F --> H[Capillary injury & increased permeability]:::action G --> I[Hypoxemia]:::urgent H --> J[Pulmonary edema & ARDS]:::urgent D --> K[RBC fragmentation on fibrin strands]:::action K --> L[Schistocytes & hemolysis]:::outcome ``` **Clinical Pearl:** The combination of **schistocytes + thrombocytopenia + respiratory failure + coagulopathy** in a cancer patient is DIC until proven otherwise. The hemolysis is **mechanical** (RBCs passing through fibrin-lined capillaries), not immune-mediated. ### Why This Is DIC, Not Other Diagnoses **Mnemonic:** **CANCER-DIC** — Consumption of platelets, Abnormal PT/aPTT, Necrosis of tissues (microthrombi), Coagulation activation, Elevated D-dimer, Respiratory distress, Disseminated fibrin deposition, Ischemia of organs, Consumption of fibrinogen. ### Pathophysiology of Pulmonary ARDS in DIC 1. **Microthrombi formation** in pulmonary capillaries → mechanical obstruction of blood flow 2. **Endothelial injury** from thrombin and fibrin deposition → increased vascular permeability 3. **Platelet and leukocyte sequestration** in lungs → inflammatory cytokine release (IL-6, TNF-α, IL-8) 4. **Surfactant dysfunction** from inflammatory mediators → alveolar collapse 5. **Result:** Severe hypoxemia (PaO₂ 58 mmHg), bilateral infiltrates, and shock **Warning:** Do NOT confuse DIC-induced ARDS with sepsis-induced ARDS. In sepsis, fibrinogen is typically **elevated** (acute phase reactant), whereas in DIC it is **consumed and low**. This patient's fibrinogen of 110 mg/dL confirms consumption, not inflammation alone. ### Management Priorities 1. **Treat the underlying cancer:** Chemotherapy (gemcitabine-based regimen for pancreatic cancer) 2. **Supportive care:** Mechanical ventilation, ICU monitoring 3. **Coagulation support:** FFP + cryoprecipitate for factor/fibrinogen replacement (target fibrinogen >100 mg/dL) 4. **Anticoagulation:** Low-dose unfractionated heparin (controversial but may reduce microthrombi formation) — use cautiously given active bleeding risk 5. **Avoid:** Platelet transfusions unless active bleeding and platelets <10,000/µL **High-Yield:** The prognosis of DIC in solid tumors is poor; mortality is 40–80% despite treatment. Early recognition and aggressive management of the underlying malignancy offer the only chance of DIC reversal. [cite:Robbins 10e Ch 13; Harrison 21e Ch 180] 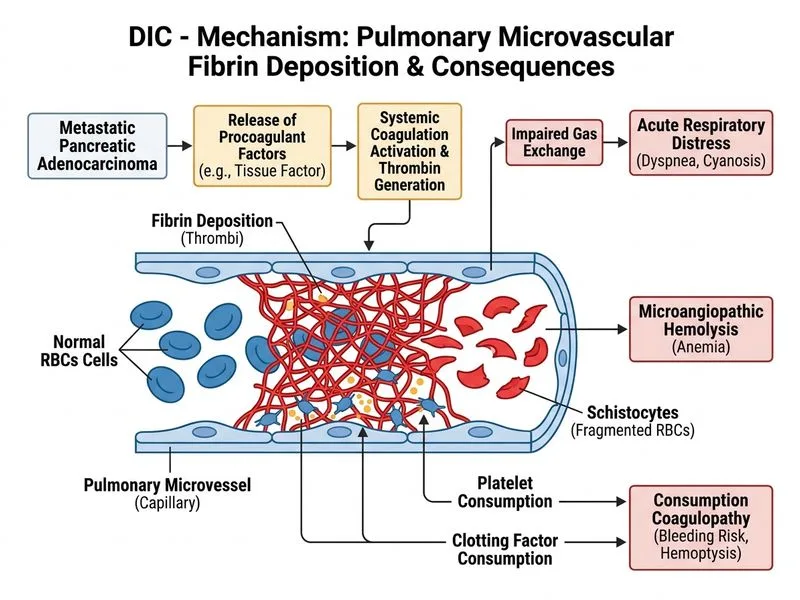
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.