## Investigation of Persistent Gestational Trophoblastic Neoplasia (PGTN) **Key Point:** When β-hCG plateaus (≥4 values over ≥3 weeks) or rises after molar evacuation, PGTN is diagnosed. The **first-line investigation** is serial serum β-hCG monitoring combined with a **chest X-ray (CXR)** — the lungs are the most common site of metastasis (~80% of metastatic GTN cases). **High-Yield (FIGO / RCOG Guidelines):** The initial workup for confirmed or suspected PGTN is: 1. **Weekly serum β-hCG** — to confirm plateau/rise and monitor response to treatment 2. **Chest X-ray** — mandatory first-line imaging to detect pulmonary metastases and assign FIGO risk score This combination (Option A) is the standard recommended first investigation before initiating chemotherapy. ### Why Not CT Abdomen/Pelvis First? CT abdomen/pelvis (Option B) is indicated **after** CXR, and only when: - CXR shows metastases (to complete staging), OR - Clinical suspicion of hepatic/splenic involvement exists CT is **not** the first-line investigation — RCOG Green-top Guideline No. 38 (2020) and FIGO 2018 explicitly recommend CXR as the initial imaging modality. CT is reserved for further staging once pulmonary disease is confirmed or when high-risk features are present. | Investigation | Role in PGTN | |---|---| | Weekly β-hCG + CXR | **First-line** — confirms PGTN, detects pulmonary mets, assigns risk score | | Transvaginal USS | Useful adjunct; assesses uterine invasion but not first-line | | CT abdomen/pelvis | Second-line; used when CXR positive or high-risk features | | MRI brain | Reserved for high-risk FIGO score or neurological symptoms | **Clinical Pearl:** The FIGO scoring system for GTN uses CXR (not CT) to count pulmonary metastases for risk stratification. A patient with only pulmonary metastases on CXR may be classified as low-risk and treated with single-agent methotrexate, whereas CT findings of hepatic metastases would upgrade to high-risk (EMA-CO regimen). **Why Other Options Are Wrong:** - **Option C (Diagnostic hysteroscopy):** Contraindicated in GTN — risk of perforation and dissemination of trophoblastic tissue. - **Option D (Serum hPL):** Not a standard marker for GTN monitoring; β-hCG remains the gold-standard tumor marker. **Reference:** RCOG Green-top Guideline No. 38 (2020); FIGO Oncology Committee Report 2018; Seckl MJ et al., *Lancet* 2010. 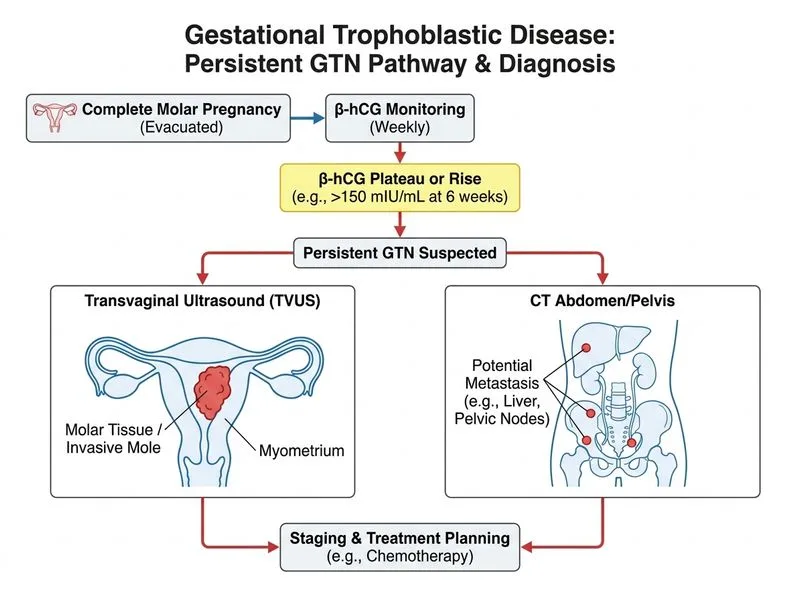
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.