## Clinical Context & Imaging Interpretation **Key Point:** The combination of acute ischemic stroke (DWI restriction in MCA territory) with **multiple scattered hypointense foci on SWI throughout the brain parenchyma** in a 72-year-old patient is most consistent with **cerebral amyloid angiopathy (CAA)**, not hypertensive arteriopathy, given the patient's age and the described distribution pattern. ## Microhemorrhages: Etiology & Imaging Patterns | Feature | Hypertensive Arteriopathy | Cerebral Amyloid Angiopathy (CAA) | Hemorrhagic Transformation (HT) | |---------|---------------------------|-----------------------------------|----------------------------------| | **Location** | Deep white matter, basal ganglia, brainstem, thalamus | Lobar (cortical/subcortical), cortical-subarachnoid | At site of acute infarct | | **Pattern** | Multiple, deep, chronic | Multiple lobar/cortical, scattered | Single or few, at infarct site | | **Age** | Middle-aged to elderly with HTN | Elderly (>70 yrs) | Any age, post-infarct | | **SWI appearance** | Hypointense foci (deep structures) | Hypointense foci (cortical/lobar, subarachnoid) | Acute hyperdensity on CT | | **Associated findings** | Lacunar infarcts, leukoaraiosis | Lobar ICH, cortical microinfarcts, superficial siderosis | Hemorrhage at infarct border | **High-Yield:** SWI is exquisitely sensitive for detecting microhemorrhages because deoxyhemoglobin and hemosiderin are paramagnetic and create local field inhomogeneities. Microhemorrhages appear as hypointense (dark) foci on SWI. ## Why This Case Points to Cerebral Amyloid Angiopathy (CAA) 1. **Age** — The patient is 72 years old; CAA is predominantly a disease of the elderly (>70 years), with amyloid-β deposition in cortical and leptomeningeal vessels 2. **Distribution** — Multiple hypointense foci **scattered throughout the brain parenchyma AND subarachnoid space** is the hallmark of CAA, reflecting cortical microhemorrhages and superficial siderosis (leptomeningeal involvement). Hypertensive arteriopathy preferentially affects deep structures (basal ganglia, thalamus, brainstem) 3. **Subarachnoid involvement** — The presence of SWI hypointensities in the subarachnoid space is characteristic of **superficial siderosis**, a recognized feature of CAA, not hypertensive arteriopathy 4. **Normal CT** — Rules out acute large-volume subarachnoid hemorrhage (which would show hyperdensity on CT); the SWI findings represent chronic microhemorrhages and superficial siderosis 5. **Atrial fibrillation context** — The acute MCA infarct is likely cardioembolic (AF not anticoagulated), while the SWI findings represent a separate, pre-existing CAA process **Boston Criteria for CAA** (modified): Multiple lobar/cortical microhemorrhages or superficial siderosis on MRI in patients ≥55 years supports probable CAA in the absence of other explanations. **Clinical Pearl:** The co-existence of cardioembolic stroke (from AF) and CAA-related microhemorrhages is clinically important because CAA significantly increases the risk of anticoagulation-related intracerebral hemorrhage. SWI findings should be carefully reviewed before initiating anticoagulation in elderly AF patients. **Why not Option C (Hypertensive Arteriopathy)?** Hypertensive arteriopathy causes microhemorrhages predominantly in **deep structures** (basal ganglia, thalamus, pons, cerebellum). The involvement of the subarachnoid space on SWI is not a feature of hypertensive small-vessel disease — it points instead to leptomeningeal amyloid deposition (CAA). [cite: Harrison's Principles of Internal Medicine, 21e, Ch. 296; Greenberg SM et al., Stroke 2022 — Updated Boston Criteria for CAA; Charidimou A et al., Nat Rev Neurol 2017] 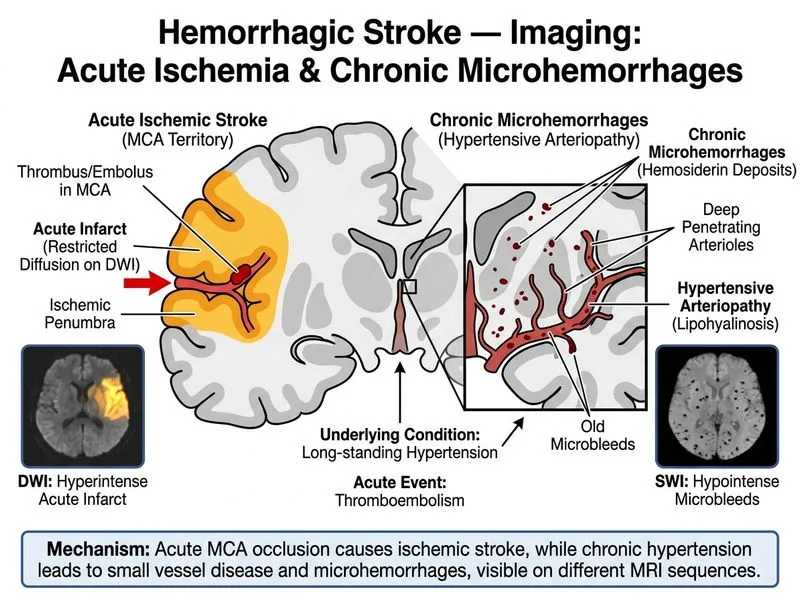
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.