## Clinical Context This patient has herpes zoster involving the V2 (maxillary) distribution of the trigeminal nerve. Although the rash is in a cranial nerve dermatome, there are no signs of ocular involvement (no mention of eye pain, photophobia, or conjunctivitis). The patient is immunocompetent (diabetes alone does not mandate IV therapy for uncomplicated zoster). ## Trigeminal Zoster: Key Considerations **Key Point:** Zoster involving the trigeminal nerve (especially V1 ophthalmic) carries risk of herpes zoster ophthalmicus (HZO) with potential for corneal scarring and vision loss. V2 and V3 involvement is lower risk but still warrants ophthalmology evaluation. **High-Yield:** Management stratification: - **V1 (ophthalmic) involvement or ocular symptoms:** Urgent ophthalmology assessment + IV acyclovir - **V2/V3 involvement WITHOUT ocular symptoms:** Oral acyclovir + planned (not urgent) ophthalmology review - **Non-cranial dermatomes:** Oral acyclovir alone; ophthalmology not needed **Clinical Pearl:** The absence of ocular symptoms (eye pain, photophobia, conjunctivitis, lid involvement) in this V2 case suggests low risk of HZO. Oral therapy is appropriate with ophthalmology follow-up to detect early signs. ## Why Oral Acyclovir + Planned Ophthalmology Review is Correct 1. **No ocular involvement:** Patient denies eye symptoms; rash is limited to skin 2. **Timing:** Day 5 of rash — still within 72-hour window for antiviral benefit (though benefit diminishes after 72 hours, early treatment is still indicated) 3. **Immunocompetence:** Diabetes alone does not require IV therapy; oral acyclovir is standard for uncomplicated zoster 4. **Ophthalmology safety net:** Planned review detects early corneal involvement or other complications ## Dosing and Duration | Parameter | Recommendation | |---|---| | **Oral acyclovir dose** | 800 mg five times daily for 7–10 days | | **Alternatives** | Valacyclovir 1000 mg TDS or famciclovir 500 mg TDS | | **Ophthalmology timing** | Within 1–2 weeks (not urgent if no ocular symptoms) | | **IV acyclovir indication** | Reserved for: disseminated zoster, immunocompromised hosts, or HZO with corneal involvement | **Mnemonic: "ZOSTER EYES"** — When to escalate to IV acyclovir in trigeminal zoster: - **Z**oster + V1 (ophthalmic) involvement - **O**cular symptoms (pain, photophobia, conjunctivitis) - **S**evere immunosuppression (HIV CD4 <200, post-transplant, malignancy on chemo) - **T**iming >72 hours but high-risk patient - **E**xtensive or disseminated rash - **R**etinal involvement (ARN in immunocompromised) **Tip:** Do not over-escalate to IV therapy in an immunocompetent patient with trigeminal zoster lacking ocular signs. Oral therapy + ophthalmology surveillance is cost-effective and evidence-based. 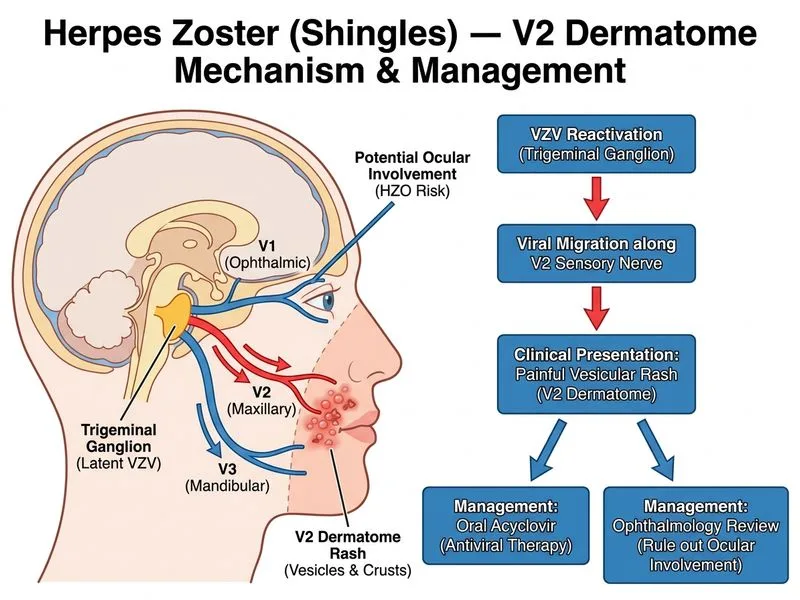
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.