## Diagnosis: Type 1 Lepra Reaction (Reversal Reaction) with Acute Neuritis ### Clinical Presentation The patient presents with: - **Borderline lepromatous leprosy** (unstable form) - **3 months into treatment** (typical onset for Type 1) - **Inflammation of existing lesions** (facial lesions reddened and painful) - **Acute neuritis** — swelling, tenderness, and loss of sensation in ulnar nerve distribution - **Afebrile** — no systemic toxicity - **Moderate bacillary load** ### Type 1 Lepra Reaction (Reversal Reaction) — Key Features **Key Point:** Type 1 lepra reaction is a **cell-mediated (Type IV hypersensitivity)** reaction occurring in unstable borderline forms (BT, BL, BB) of leprosy, typically within the first year of treatment. It is characterized by inflammation of *existing* lesions and **acute neuritis**. **High-Yield:** Type 1 lepra reaction is the **most common cause of acute neuritis in leprosy patients**. Neuritis can occur in isolation (neuritis-only reaction) or with skin lesion inflammation. The reaction reflects **upgrading or downgrading** of immunity as the immune system responds to treatment. ### Pathogenesis Type 1 reaction results from **delayed-type hypersensitivity** to *Mycobacterium leprae* antigens. As immune function improves with treatment, T-cell-mediated immunity increases, causing inflammation of lesions and nerves containing bacilli. This can manifest as: - **Upgrading reaction** — shift toward tuberculoid (TT) pole (better prognosis) - **Downgrading reaction** — shift toward lepromatous (LL) pole (worse prognosis) ### Neuritis in Type 1 Lepra Reaction **Clinical Pearl:** Acute neuritis in Type 1 lepra reaction is a medical emergency. Rapid inflammation within the nerve sheath can cause: 1. **Sudden loss of sensation** (as in this case — ulnar nerve, 4th–5th fingers) 2. **Motor weakness** — wrist drop (radial), foot drop (common peroneal) 3. **Permanent nerve damage** if not treated promptly **Mnemonic: SILENT — Sudden Inflammatory Lesion Enlargement and Neuritis in Type 1 Reaction** ### Differential: Type 1 vs Type 2 Lepra Reaction | Feature | Type 1 (Reversal Reaction) | Type 2 (ENL) | |---------|---------------------------|---------------| | **Mechanism** | Cell-mediated (Type IV) | Immune complex (Type III) | | **Leprosy type** | BT, BL, BB (unstable) | LL, BL (high bacillary load) | | **Onset** | Within 1 year of treatment | 6 months to 2 years | | **Skin lesions** | Existing lesions inflamed | NEW painful nodules | | **Neuritis** | Common, acute, prominent | Rare | | **Systemic signs** | Absent or mild | Fever, malaise, lymphadenopathy | | **Temperature** | Normal | Elevated | | **Treatment** | Corticosteroids (high-dose, urgent) | NSAIDs, corticosteroids, thalidomide | ### Management of Type 1 Lepra Reaction with Neuritis **Urgent action required:** 1. **High-dose corticosteroids** — Prednisolone 1–2 mg/kg/day (e.g., 80–100 mg/day), taper over 8–12 weeks 2. **Continue MDT** — Do NOT stop antileprosy drugs 3. **NSAIDs** — For symptomatic relief 4. **Nerve care** — Splinting, physiotherapy to prevent contractures 5. **Monitor for complications** — Permanent sensory/motor loss if treatment delayed **Warning:** Delay in starting high-dose corticosteroids can result in permanent nerve damage. This is one of the few situations in leprosy where corticosteroids are started *immediately* without waiting for investigations. ### Why Thalidomide is NOT First-Line for Type 1 **High-Yield:** Thalidomide is the drug of choice for **Type 2 lepra reaction (ENL)**, not Type 1. In Type 1 reactions, **corticosteroids are the mainstay** of treatment. Thalidomide has no role in Type 1 lepra reaction. ### Prognosis - **Upgrading reaction** — indicates improving immunity; good long-term prognosis - **Downgrading reaction** — indicates worsening immunity; poorer prognosis - **Neuritis outcome** — depends on promptness of treatment; early high-dose corticosteroids prevent permanent disability 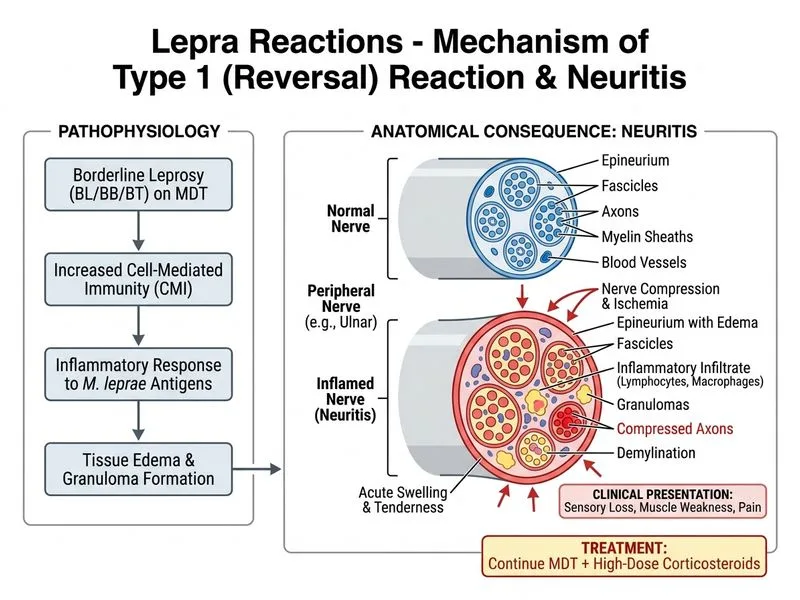
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.