## Investigation of Choice for Peripheral Lung Nodule Diagnosis ### Clinical Context The patient has a **peripheral lung nodule** (4 cm, right lower lobe) suspicious for malignancy on imaging. The goal is to obtain tissue for histological diagnosis to confirm lung cancer and determine cell type (adenocarcinoma, squamous cell, etc.). ### Why CT-Guided Percutaneous Needle Biopsy? **Key Point:** CT-guided percutaneous needle biopsy is the **gold standard for diagnosis of peripheral lung nodules** >2 cm that are accessible and not in close proximity to vital structures. **High-Yield:** This technique offers: 1. **Direct visualization** of the nodule under CT guidance 2. **High diagnostic yield** (>90% for nodules >2 cm) 3. **Tissue procurement** — allows core biopsy for histology, immunohistochemistry, and molecular testing (EGFR, ALK, PD-L1) 4. **Minimal morbidity** — outpatient procedure with low complication rate 5. **Rapid diagnosis** — results available within days ### Comparison with Other Investigations | Investigation | Indication | Yield | Limitation | |---|---|---|---| | **CT-guided needle biopsy** | Peripheral nodules >2 cm, accessible | >90% | Requires radiologist expertise | | TTNAC | Peripheral nodules, high-risk patients | 85–95% | Cytology only; may not subtype | | Bronchoscopy | Central lesions, endobronchial disease | 40–60% for peripheral | Poor yield for peripheral nodules | | Sputum cytology | Screening, central airway lesions | <50% | Very low sensitivity for nodules | **Clinical Pearl:** TTNAC is an acceptable alternative if the nodule is >2 cm and the patient is a poor surgical candidate, but **core biopsy (needle biopsy) is preferred** because it provides tissue architecture and allows molecular profiling. ### Molecular Testing Requirement **Key Point:** In non-small cell lung cancer (NSCLC), tissue diagnosis is essential not only for histology but also for **predictive molecular testing**: - EGFR mutations (adenocarcinoma) - ALK rearrangements (adenocarcinoma) - PD-L1 expression (immunotherapy eligibility) - KRAS mutations (adenocarcinoma) Cytology specimens are often insufficient for these assays; **core tissue is mandatory**. [cite:Robbins 10e Ch 15] 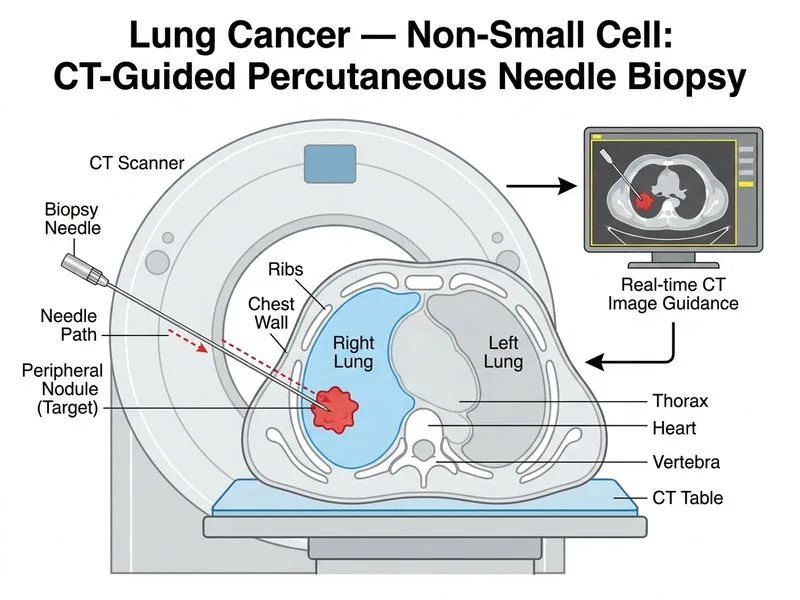
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.