## TNM Staging & SCLC Classification This patient has **locally advanced SCLC** (Stage IIIA) based on: - **T3** tumor (invasion into main bronchus) - **N2** disease (ipsilateral mediastinal lymph node involvement, stations 4L and 7) - **M0** (no distant metastases documented) **Key Point:** SCLC staging in clinical practice often uses the **two-tier Veterans Administration Lung Study Group (VALSG) classification**: - **Limited-stage disease (LD-SCLC):** Tumor confined to one hemithorax and regional lymph nodes (TNM stages I–IIIA) - **Extensive-stage disease (ED-SCLC):** Distant metastases or contralateral involvement (TNM stage IIIB–IV) This patient has **limited-stage disease** (T3N2M0 = Stage IIIA). ## Treatment Algorithm for LD-SCLC ```mermaid flowchart TD A[Limited-Stage SCLC]:::outcome --> B{Performance Status 0-1?}:::decision B -->|Yes| C[Concurrent Chemoradiotherapy]:::action B -->|No| D[Sequential CRT or Palliative Tx]:::action C --> E[Platinum-Etoposide x 4-6 cycles]:::action E --> F[Concurrent Thoracic RT 45 Gy/30 fx]:::action F --> G[Prophylactic Cranial Irradiation if CR]:::action G --> H[Median OS 20-24 months]:::outcome D --> I[Chemotherapy first, then RT]:::action I --> J[Median OS 12-15 months]:::outcome ``` ## Standard Treatment Regimen **High-Yield:** The **gold standard for LD-SCLC** is: 1. **Concurrent chemoradiotherapy (concurrent CRT)** — superior to sequential CRT - **Chemotherapy:** Cisplatin 60 mg/m² day 1 + Etoposide 120 mg/m² days 1–3, repeated every 3 weeks × 4–6 cycles - **Thoracic radiotherapy:** 45 Gy in 30 fractions (1.5 Gy twice daily), starting with cycle 1 or 2 2. **Prophylactic cranial irradiation (PCI):** 25 Gy in 10 fractions if complete response (CR) achieved - Reduces brain metastasis incidence from ~40% to ~15% - Improves 2-year overall survival by ~5% **Clinical Pearl:** Concurrent CRT achieves: - **Complete response rate:** 50–60% - **Median overall survival:** 20–24 months (vs. 12–15 months with sequential CRT) - **2-year survival:** ~30–35% ## Why Concurrent CRT? | Feature | Concurrent CRT | Sequential CRT | |---------|---|---| | **Median OS** | 20–24 months | 12–15 months | | **2-yr survival** | 30–35% | 15–20% | | **Toxicity** | Higher (esophagitis, pneumonitis) | Lower | | **Compliance** | Demanding (6–8 weeks) | More flexible | | **Indication** | PS 0–1, LD-SCLC | PS ≥2 or poor tolerance | **Mnemonic: LD-SCLC Treatment** — **"Concurrent Chemo-RT + PCI"** — Limited-stage SCLC is curable with aggressive concurrent therapy; always add PCI if CR achieved. [cite:Harrison 21e Ch 297; Robbins 10e Ch 15] 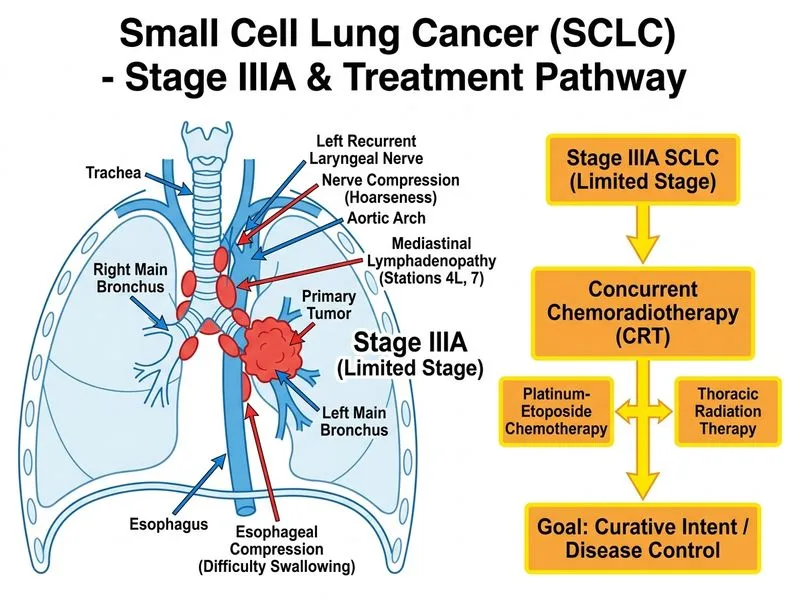
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.