## Classification and MDT Selection This patient has **tuberculoid leprosy (TT)** based on: - Few well-demarcated lesions (≤5) - Diminished sensation (asymmetric nerve involvement) - Low bacterial load (2+ AFB on slit-skin smear) - Clear demarcation and asymmetry ## WHO MDT Regimen for Tuberculoid Leprosy **Key Point:** Tuberculoid leprosy is classified as **paucibacillary (PB)** disease and requires a 6-month MDT regimen. ### Standard MDT-PB Regimen | Drug | Dose | Frequency | Duration | |------|------|-----------|----------| | Rifampicin | 600 mg | Monthly supervised + daily unsupervised | 6 months | | Dapsone | 100 mg | Daily (unsupervised) | 6 months | | Clofazimine | 300 mg (monthly) + 50 mg (daily) | Mixed schedule | 6 months | **High-Yield:** The MDT-PB regimen consists of **rifampicin + dapsone + clofazimine for 6 months**, with 12 supervised doses of rifampicin and 12 supervised doses of clofazimine. ## Rationale for This Regimen **Clinical Pearl:** Clofazimine is included even in tuberculoid leprosy to: - Provide anti-inflammatory action (reduces type 1 reactions) - Cover any undetected borderline lesions - Ensure bactericidal coverage **Mnemonic:** **RDC** = **R**ifampicin (backbone), **D**apsone (folate antagonist), **C**lofazimine (lipophilic, anti-inflammatory) ## Why Duration is 6 Months, Not 12 or 24 1. **Bacteriological clearance:** Rifampicin renders patients non-infectious within 2 weeks 2. **Lesion resolution:** Most lesions show significant improvement by 6 months 3. **WHO guidelines:** 6-month MDT-PB is the standard for all paucibacillary forms (TT, BT, and indeterminate) 4. **Relapse rates:** <1% relapse rate with 6-month MDT-PB when properly supervised **Warning:** Extending MDT to 12 or 24 months does NOT improve outcomes and increases toxicity risk (especially dapsone-induced hemolysis and clofazimine pigmentation). 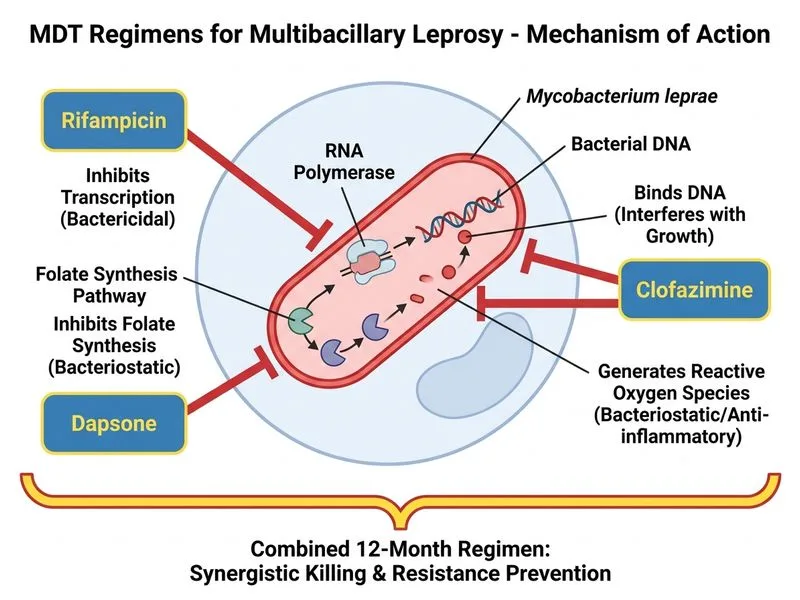
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.