## Clinical Staging and Management Decision ### Breslow Thickness and Stage Classification **Key Point:** A Breslow thickness of 2.8 mm (T3a — no ulceration, mitotic rate ≥1/mm²) with negative sentinel lymph node biopsy classifies this melanoma as **Stage IIB** per AJCC 8th Edition. High mitotic rate (3/mm²) and Clark level IV confer significant recurrence risk. | Breslow Thickness | Ulceration | Stage (Node-negative) | |---|---|---| | 1.0–2.0 mm | Absent | IIA | | 1.0–2.0 mm | Present | IIB | | 2.0–4.0 mm | Absent | IIB | | 2.0–4.0 mm | Present | IIC | | >4.0 mm | Absent | IIC | ### Best Next Step: Staging + Adjuvant Immunotherapy **High-Yield:** Per **NCCN Melanoma Guidelines (2023–2024)** and **ASCO guidelines**, the recommended management for resected Stage IIB–IIC melanoma is: 1. **Whole-body imaging (PET-CT or CT chest/abdomen/pelvis ± brain MRI)** to exclude occult distant metastases before initiating adjuvant therapy. 2. **Adjuvant checkpoint inhibitor immunotherapy** — **pembrolizumab** (KEYNOTE-716 trial) is FDA-approved for Stage IIB/IIC resected melanoma; **nivolumab** is approved for Stage III. These agents have replaced interferon-alpha as the standard of care due to superior recurrence-free survival and tolerability. **Clinical Pearl:** The KEYNOTE-716 trial (NEJM 2022) demonstrated that adjuvant pembrolizumab significantly improved recurrence-free survival in Stage IIB/IIC melanoma vs. placebo, establishing immunotherapy as first-line adjuvant treatment. Interferon-alpha is now considered second-line or historical standard only. [cite: NCCN Melanoma v2.2024; KEYNOTE-716, NEJM 2022] ### Surgical Adequacy **Key Point:** The initial excision was performed with only 5 mm margins. For Breslow thickness 2.0–4.0 mm, guidelines recommend **2 cm margins** (NCCN, BAD guidelines). Therefore, **re-excision (wide local excision) with 2 cm margins** is also required — however, this is already incorporated into the standard workflow *before* adjuvant therapy. Option A correctly sequences staging imaging → adjuvant immunotherapy, which implicitly follows adequate surgical re-excision as part of the complete management plan. --- ## Why Each Option Is Correct or Incorrect **Option A (Correct):** PET-CT to exclude distant metastases followed by adjuvant immunotherapy (pembrolizumab/nivolumab) is the current evidence-based standard of care for Stage IIB resected melanoma per NCCN 2023–2024 guidelines. **Option B (Observation only):** Insufficient for Stage IIB melanoma. The high Breslow thickness (2.8 mm), Clark level IV, and mitotic rate (3/mm²) confer >20% 10-year mortality risk, warranting adjuvant systemic therapy. **Option C (Wide local excision + interferon-alpha):** While re-excision to 2 cm margins is necessary, adjuvant interferon-alpha is **no longer the standard of care**. It has been superseded by checkpoint inhibitors with superior efficacy and tolerability. Recommending interferon-alpha as the primary adjuvant agent is inconsistent with current NCCN/ASCO guidelines (2023–2024). **Option D (Completion lymphadenectomy):** Completion lymphadenectomy is **not indicated** for SLNB-negative patients. The MSLT-II trial confirmed no overall survival benefit and increased morbidity (lymphedema). Reserved for SLNB-positive disease only. 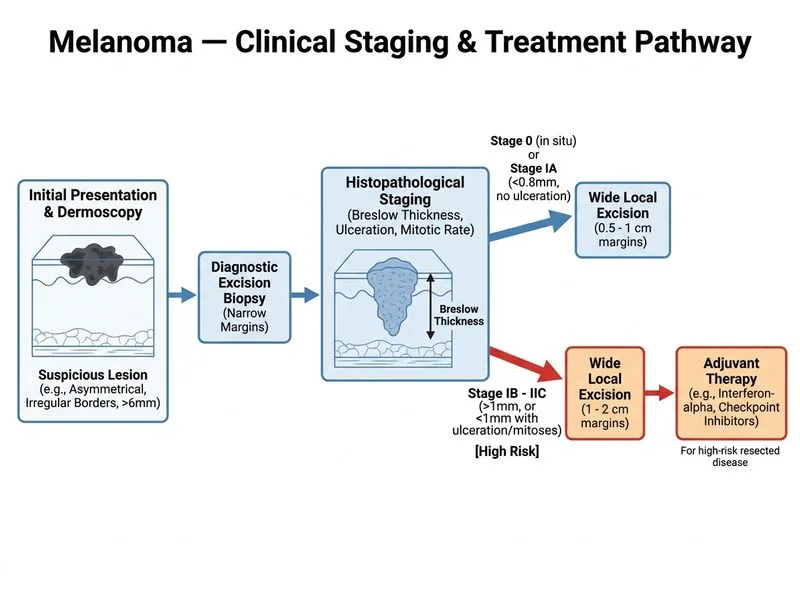
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.