## Clinical Context This patient has **stage IIIB melanoma** (Breslow thickness 3.2 mm with ulceration, positive sentinel lymph node with micrometastases). The presence of nodal involvement mandates **completion lymphadenectomy** as the next surgical step. ## Staging Classification ```mermaid flowchart TD A[Melanoma with positive SLNB]:::outcome --> B{Extent of nodal disease?}:::decision B -->|Micrometastasis in 1 node| C[Stage IIIA or IIIB]:::outcome B -->|Macrometastasis or multiple nodes| D[Stage IIIB or IIIC]:::outcome C --> E{Breslow thickness & ulceration?}:::decision E -->|Breslow ≤2 mm, no ulc| F[Stage IIIA]:::outcome E -->|Breslow > 2 mm OR ulcerated| G[Stage IIIB]:::outcome G --> H[Completion lymphadenectomy]:::action H --> I[Consider adjuvant therapy]:::action ``` ## Key Point: **Completion lymphadenectomy is the standard of care for patients with positive sentinel lymph node biopsy (stage III melanoma).** This involves removal of all lymph nodes in the involved regional basin (in this case, right inguinal lymph nodes). This is both **therapeutic** (removes involved nodes) and **prognostic** (determines final staging and guides adjuvant therapy decisions). ## High-Yield: - **Positive SLNB = Stage III disease** (regional nodal involvement). - **Completion lymphadenectomy is mandatory** in all SLNB-positive patients unless the patient is unfit or declines surgery. - Micrometastases (tumour cells <0.1 mm or <200 cells) vs. macrometastases (>0.1 mm) affect substaging but both require completion lymphadenectomy. - **Adjuvant therapy (interferon-α, checkpoint inhibitors like nivolumab/pembrolizumab, or targeted therapy) is considered AFTER lymphadenectomy** based on final nodal burden and other risk factors. ## Clinical Pearl: **Nodular melanoma** is an aggressive subtype with: - Vertical growth phase from onset (no radial growth phase). - Higher Breslow thickness at diagnosis (often >2 mm). - Worse prognosis than superficial spreading melanoma. - Ulceration is common and indicates poor prognosis. ## Adjuvant Therapy Considerations (Post-Lymphadenectomy) | Indication | Preferred Agent | |------------|----------------| | Stage IIIB–IIIC (node-positive) | Checkpoint inhibitor (nivolumab, pembrolizumab) or interferon-α | | BRAF-mutated stage III | Dabrafenib + trametinib (targeted therapy) | | High-risk stage II | Interferon-α or checkpoint inhibitor (if ulcerated or high mitotic rate) | **Dacarbazine monotherapy is outdated** and has been largely replaced by checkpoint inhibitors and targeted therapies, which offer superior outcomes. 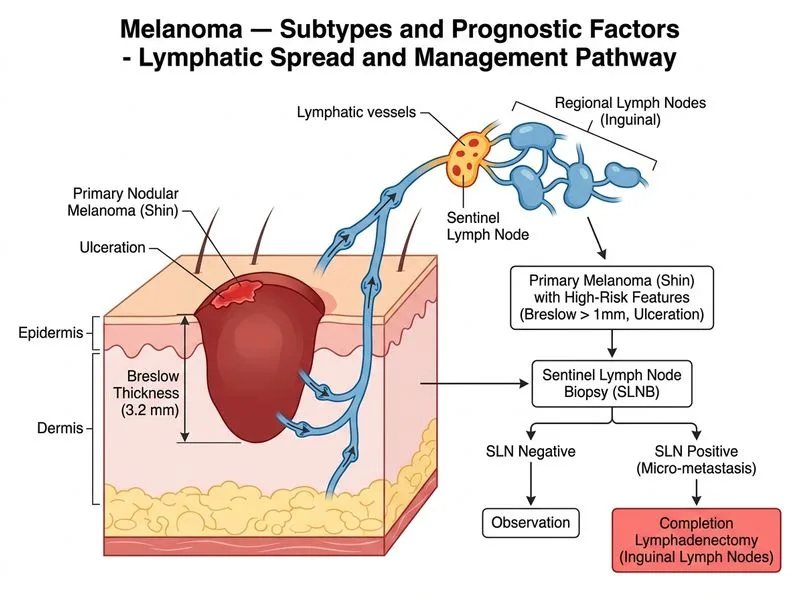
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.