## Management of Locally Advanced Nasopharyngeal Carcinoma (T3N2M0) ### Staging Context **Key Point:** T3N2M0 represents locally advanced disease with nodal involvement — this is a non-metastatic but regionally advanced tumour that requires multimodal therapy. ### Why Concurrent Chemoradiotherapy (CCRT) is the Standard of Care **High-Yield:** For nasopharyngeal carcinoma (NPC), especially in endemic regions (Southeast Asia, Southern China, North Africa), concurrent chemoradiotherapy with cisplatin is the gold-standard treatment for stages II–IVA (non-metastatic locally advanced disease). **Clinical Pearl:** NPC has unique epidemiology and biology: - Strong association with EBV (especially in endemic areas) - High radiosensitivity compared to other head-and-neck cancers - Propensity for early nodal involvement - Surgery has limited role (nasopharynx is difficult to access; high morbidity) ### Evidence Base | Parameter | CCRT | RT Alone | Surgery | Palliative CTx | |-----------|------|----------|---------|----------------| | **5-year OS (Stage III–IVA)** | 60–75% | 40–50% | Not applicable | <20% | | **Locoregional control** | Excellent | Fair | Not primary | Poor | | **Toxicity** | Moderate (manageable) | Lower acute, higher late | High morbidity | Variable | | **Indication** | Locally advanced (T3–T4, any N) | Early stage (T1–T2, N0) | Salvage only (recurrent) | Metastatic/unfit | **Key Point:** Cisplatin-based CCRT improves overall survival by ~10–15% compared to RT alone in locally advanced NPC. ### Treatment Algorithm for T3N2M0 NPC ```mermaid flowchart TD A[Nasopharyngeal Carcinoma Diagnosed]:::outcome --> B{Metastatic?}:::decision B -->|Yes| C[Palliative chemotherapy]:::action B -->|No| D{Stage?}:::decision D -->|Early Stage<br/>T1-T2, N0| E[Radiotherapy alone<br/>70 Gy]:::action D -->|Locally Advanced<br/>T3-T4 or N1-N3| F[Concurrent Chemoradiotherapy<br/>Cisplatin 100 mg/m² Q3W]:::action F --> G[Induction chemotherapy<br/>optional for N3]:::action F --> H[Radiotherapy 70 Gy<br/>concurrent with Cisplatin]:::action H --> I{Complete response?}:::decision I -->|Yes| J[Adjuvant chemotherapy<br/>optional]:::action I -->|No| K[Salvage surgery/<br/>Re-irradiation]:::action ``` ### Cisplatin Dosing in CCRT - **Standard:** 100 mg/m² IV on days 1, 22, 43 of radiotherapy (3 cycles) - **Alternative:** 40 mg/m² weekly × 7 cycles (for renal impairment) - **Monitoring:** Renal function, hearing, neuropathy before each cycle **Warning:** Cisplatin is contraindicated if creatinine clearance <60 mL/min or significant hearing loss; carboplatin may be substituted. ### Why Other Options Are Not Optimal for T3N2M0 1. **Surgery alone:** Nasopharynx is surgically inaccessible without significant morbidity; endoscopic resection has no role in primary treatment. 2. **RT alone:** Insufficient locoregional control in advanced nodal disease; survival inferior to CCRT. 3. **Palliative chemotherapy:** Reserved for metastatic (M1) disease or unfit patients; not appropriate for fit patient with non-metastatic advanced disease. **Clinical Pearl:** Induction chemotherapy (platinum + 5-FU) may be added before CCRT in N3 disease, though this remains debated. [cite:Harrison 21e Ch 82] 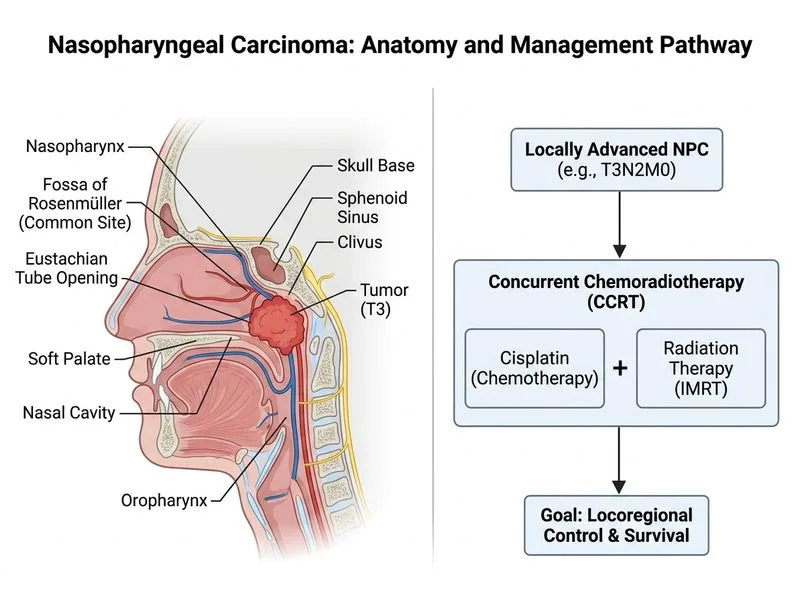
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.