## Enzyme Inhibition in Organophosphate Poisoning **Key Point:** Organophosphate compounds form a covalent phosphodiester bond with the serine residue in the active site of acetylcholinesterase (AChE), causing irreversible inhibition. ### Mechanism of Action Organophosphates phosphorylate the hydroxyl group of serine-203 in the esterase site of AChE. Unlike the reversible inhibition seen with carbamates, this phosphorylated enzyme undergoes **"aging"** — a process where the alkyl group is lost, making the inhibition essentially irreversible within the clinical timeframe. ### Biochemical Consequence | Feature | Normal | With OP Poisoning | | --- | --- | --- | | AChE activity | 100% | <20% (severe cases) | | Acetylcholine (ACh) at NMJ | Rapidly hydrolyzed | Accumulates | | Cholinergic overstimulation | Absent | Marked (muscarinic + nicotinic) | | Recovery timeline | N/A | Days to weeks (new enzyme synthesis) | **High-Yield:** The irreversible nature of OP-AChE inhibition distinguishes it from carbamate poisoning, which is reversible. This has forensic and therapeutic implications — atropine and pralidoxime (2-PAM) are effective only if given early, before aging occurs. **Clinical Pearl:** Butyrylcholinesterase (plasma cholinesterase) is also inhibited by OP compounds, but its inhibition is less clinically significant than AChE inhibition at the neuromuscular junction and autonomic synapses. ## Why AChE is the Target AChE is the primary enzyme responsible for terminating cholinergic neurotransmission at: - Neuromuscular junctions (skeletal muscle) - Autonomic ganglia - Parasympathetic postganglionic synapses - Central nervous system cholinergic synapses Inhibition at all these sites produces the characteristic muscarinic and nicotinic signs of OP poisoning. 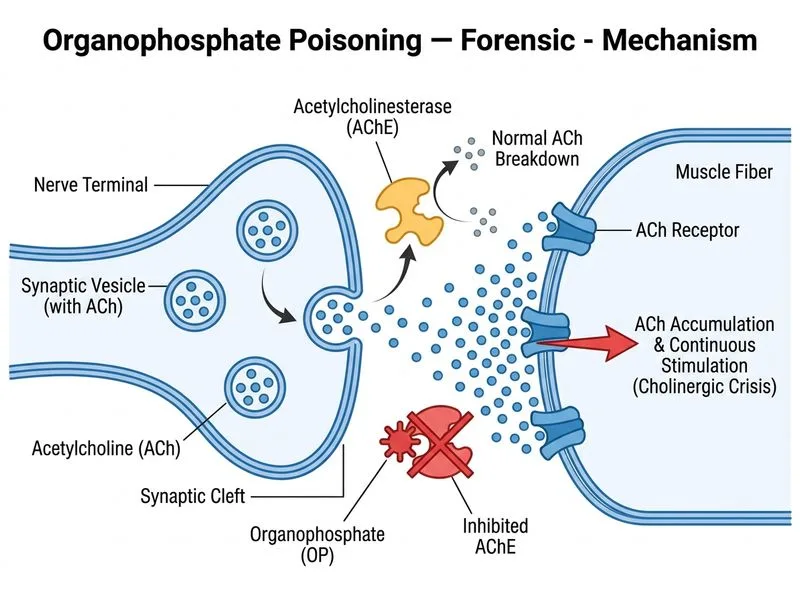
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.