## Maintenance Therapy in Epithelial Ovarian Cancer: HRD-Negative Disease **Key Point:** In patients with **HRD-negative, BRCA wild-type epithelial ovarian cancer** who achieve complete response to platinum–taxane chemotherapy, **bevacizumab monotherapy** is the most evidence-based maintenance strategy. PARP inhibitors are reserved for HRD-positive or BRCA-mutated tumors. ### Maintenance Therapy Algorithm by Molecular Status ```mermaid flowchart TD A[Epithelial Ovarian Cancer<br/>Complete Response to Chemo]:::outcome A --> B{BRCA-mutated<br/>or HRD-positive?}:::decision B -->|Yes| C[PARP inhibitor<br/>maintenance<br/>3 years]:::action B -->|No| D{High-risk<br/>features?}:::decision D -->|Yes<br/>Stage III/IV| E[Bevacizumab<br/>15 months]:::action D -->|No<br/>Early stage| F[Observation or<br/>bevacizumab<br/>per PARP trial]:::action C --> G[Superior PFS<br/>and OS vs observation]:::outcome E --> H[Improved PFS<br/>vs observation]:::outcome ``` ### Maintenance Therapy Comparison by Molecular Status | Molecular Status | Recommended Maintenance | Evidence | Duration | Benefit | |------------------|------------------------|----------|----------|----------| | **BRCA-mutated (germline or somatic)** | PARP inhibitor (olaparib, rucaparib) | SOLO2, ARIEL3 | 3 years | OS benefit (median ~3 years PFS gain) | | **HRD-positive (BRCA-WT)** | PARP inhibitor | SOLO1, ARIEL3 | 3 years | OS benefit (~2 years PFS gain) | | **HRD-negative, BRCA-WT (THIS CASE)** | **Bevacizumab monotherapy** | GOG-218, ICON7 | 15 months | PFS improvement (~3–4 months); OS benefit in some subgroups | | **HRD-negative, early stage** | Observation or bevacizumab | ICON7 subgroup | — | Bevacizumab may offer modest benefit | **High-Yield:** The **SOLO1 trial** (PARP inhibitor olaparib in BRCA-mutated) and **ARIEL3 trial** (PARP inhibitor rucaparib in HRD-positive) showed dramatic OS benefits. However, **PARP inhibitors are NOT indicated in HRD-negative disease** because the homologous recombination repair deficiency is the mechanism of PARP sensitivity. ### Why This Patient Gets Bevacizumab, Not PARP Inhibitor **Clinical Pearl:** This patient has: - **BRCA1 wild-type** → no germline BRCA mutation - **Low HRD score** → no somatic homologous recombination deficiency - **Stage IIB** (high-risk) → benefits from bevacizumab per GOG-218 and ICON7 Bevacizumab (anti-VEGF) is the standard of care in this molecular and clinical context. It improves progression-free survival by ~3–4 months and may provide overall survival benefit in Stage III/IV disease. **Key Point:** PARP inhibitors work by **synthetic lethality** in HRD-positive cells (BRCA-mutated or somatic HR-deficient). In HRD-negative tumors, the cells retain intact homologous recombination capacity and can repair PARP-induced DNA damage, rendering PARP inhibitors ineffective. ### Treatment Timeline for This Patient 1. **Carboplatin–paclitaxel** (6 cycles) → Complete response ✓ 2. **Bevacizumab maintenance** (15 months) → Improves PFS 3. **Surveillance** → CA-125, imaging, clinical exam every 3 months 4. **Recurrence management** → Platinum rechallenge ± bevacizumab if platinum-sensitive **Mnemonic:** **HRD-PARP rule**: - **H**RD-positive or **B**RCA-mutated → **PARP** inhibitor - **H**RD-negative → **B**evacizumab (or observation) 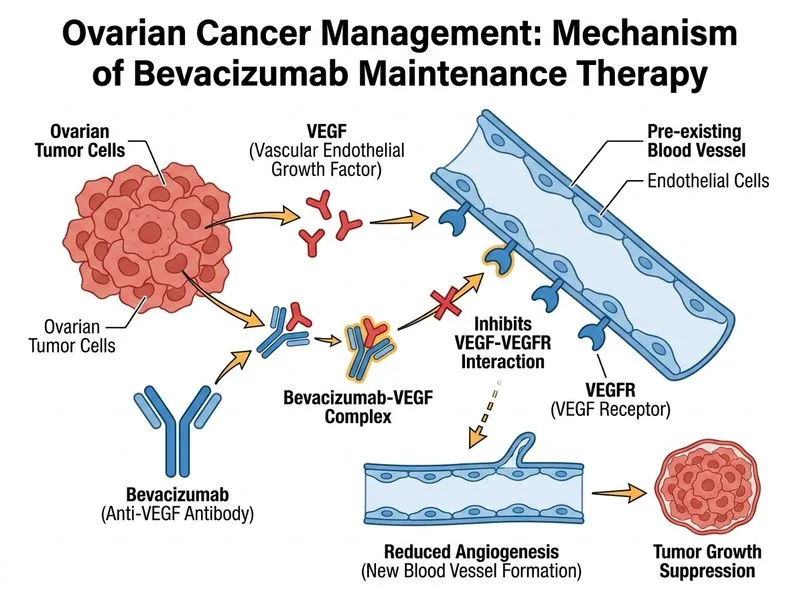
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.