## First-Line Management of Stage IIIC High-Grade Serous Ovarian Cancer ### Clinical Context This patient has: - **Stage IIIC disease** (peritoneal implants >2 cm on diaphragm and pelvic peritoneum) - **High-grade serous histology** (most aggressive epithelial subtype) - **Primary cytoreductive surgery already completed** (TAH + BSO + staging laparotomy performed) - **Normal renal function** (eligible for platinum-based therapy with no contraindications) ### Current Standard of Care **Key Point:** For Stage III–IV high-grade serous ovarian cancer where **primary debulking surgery has been performed**, the standard first-line regimen is **carboplatin + paclitaxel × 6 cycles FOLLOWED BY bevacizumab maintenance for 15 months** (per NCCN 2023 and ESMO guidelines). ### Evidence Base: GOG-218 and ICON7 Trials | Trial | Patient Population | Intervention | Primary Outcome | | --- | --- | --- | --- | | **GOG-218** | Stage III–IV, primarily **optimally debulked** | Carboplatin/paclitaxel ± bevacizumab concurrent + maintenance | Bevacizumab maintenance arm: PFS improved by ~3.8 months vs. chemotherapy alone | | **ICON7** | Stage III–IV (including high-risk suboptimal) | Carboplatin/paclitaxel ± bevacizumab | OS benefit demonstrated specifically in **high-risk subgroup** (Stage IIIC/IV or suboptimal debulking) | **High-Yield:** GOG-218 enrolled predominantly optimally debulked patients and demonstrated PFS benefit with bevacizumab maintenance. ICON7 confirmed OS benefit in high-risk patients (Stage IIIC qualifies). The 15-month maintenance duration is the standard cited in NCCN guidelines (some ESMO references cite 12–15 months; for NEET PG purposes, 15 months is the accepted answer). ### Why This Regimen? 1. **Carboplatin + Paclitaxel:** Gold standard since GOG-111 (1996). Platinum-taxane combination is superior to either drug alone or carboplatin monotherapy. 2. **6 cycles:** Standard duration; no benefit from extending beyond 6 cycles in optimally debulked patients. 3. **Bevacizumab maintenance (15 months):** Anti-VEGF monoclonal antibody that inhibits tumor angiogenesis. Adds ~4–6 months PFS benefit and improves OS in high-risk subgroups. Stage IIIC disease qualifies as high-risk. ### Why Not the Other Options? - **Option A (Carboplatin + paclitaxel alone, no maintenance):** Outdated for Stage IIIC high-grade serous OC. Current guidelines recommend bevacizumab maintenance for Stage III–IV patients after chemotherapy completion. - **Option B (Neoadjuvant chemotherapy → interval debulking):** This approach is reserved for patients in whom **primary optimal debulking is not feasible** (e.g., unresectable disease, poor performance status, or high surgical risk). In this vignette, primary surgery has **already been completed**; neoadjuvant therapy is therefore not applicable. - **Option C (Carboplatin monotherapy):** Substandard. Platinum-taxane combination is superior. Monotherapy is reserved for patients who cannot tolerate paclitaxel (e.g., severe pre-existing neuropathy or hypersensitivity). **Clinical Pearl:** Bevacizumab is contraindicated in uncontrolled hypertension, recent arterial thromboembolism, bowel perforation risk, or significant renal impairment. This patient has normal renal function and no stated contraindications, making bevacizumab maintenance appropriate. **Mnemonic:** **BEVO** = **B**evacizumab for **E**pithelial o**V**arian cancer maintenance in Stage III–**IV** (post-primary surgery + chemotherapy). [cite: NCCN Ovarian Cancer Guidelines v1.2023; GOG-218 (Burger et al., NEJM 2011); ICON7 (Perren et al., NEJM 2011); KD Tripathi Essentials of Medical Pharmacology, 8th ed.] 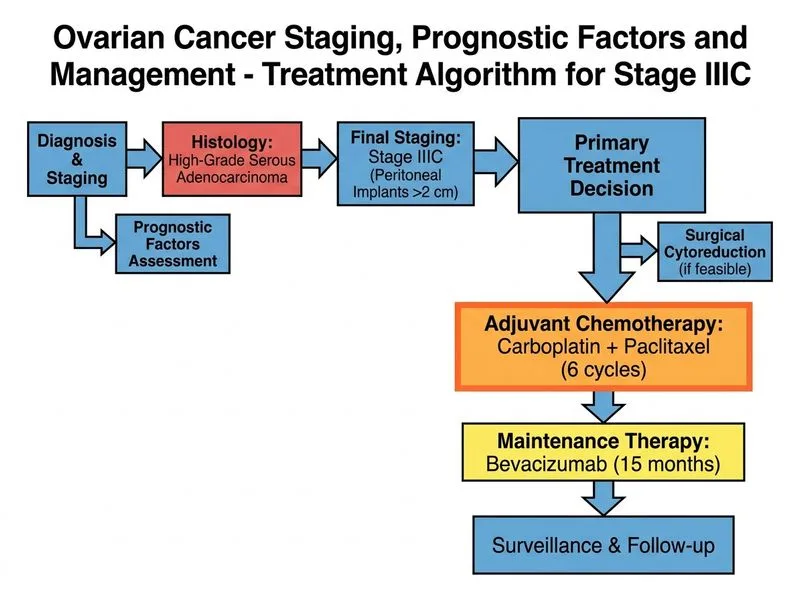
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.