## Treatment of Pemphigus Vulgaris ### First-Line Therapy **High-Yield:** Systemic corticosteroids are the gold standard initial treatment for pemphigus vulgaris. High-dose oral corticosteroids (prednisolone 1–1.5 mg/kg/day) are required to suppress autoantibody production and achieve disease control. **Key Point:** Topical corticosteroids and antihistamines alone are insufficient for systemic pemphigus vulgaris. They may be used as adjuncts for oral lesions but cannot control the underlying autoimmune process. ### Treatment Algorithm ```mermaid flowchart TD A[Pemphigus Vulgaris Diagnosis]:::outcome --> B[Initiate systemic corticosteroids]:::action B --> C[Prednisolone 1-1.5 mg/kg/day]:::action C --> D{Response in 1-2 weeks?}:::decision D -->|Yes| E[Gradual taper over months]:::action D -->|No| F[Add steroid-sparing agent]:::action F --> G[Azathioprine or Mycophenolate]:::action E --> H[Maintenance therapy]:::action H --> I[Prednisolone 0.5 mg/kg/day or less]:::action ``` ### Steroid-Sparing Agents **Mnemonic: AMMO** (Azathioprine, Mycophenolate, Methotrexate, Others) | Agent | Mechanism | Onset | Typical Dose | |-------|-----------|-------|-------------| | **Azathioprine** | Purine antagonist; inhibits T-cell proliferation | 6–8 weeks | 1–2 mg/kg/day | | **Mycophenolate mofetil** | Selective IMPDH inhibitor; inhibits B-cell proliferation | 4–6 weeks | 1–3 g/day | | **Methotrexate** | Folate antagonist; immunosuppression | 4–6 weeks | 15–25 mg/week | | **Rituximab** | Anti-CD20 monoclonal antibody; B-cell depletion | 2–4 weeks | 375 mg/m² weekly × 4 | **Clinical Pearl:** Rituximab is increasingly used in severe or refractory pemphigus vulgaris and has shown excellent outcomes in clinical trials, but prednisolone remains the initial therapy. ### Rationale for Systemic Corticosteroids 1. **Rapid anti-inflammatory effect:** Suppresses T-cell and B-cell function 2. **Reduces autoantibody production:** Targets the underlying pathogenic mechanism 3. **Achieves remission:** 80–90% of patients achieve clinical remission within 4–8 weeks 4. **Prevents complications:** Prevents secondary infection of erosions and systemic complications **Warning:** Abrupt withdrawal of corticosteroids can precipitate disease flare. Gradual tapering over months is essential. ### Monitoring During Treatment - **Clinical response:** Cessation of new blister formation (1–2 weeks); healing of erosions (2–4 weeks) - **Serological response:** Decline in anti-Dsg3 titers (correlates with clinical improvement) - **Adverse effects:** Screen for hyperglycemia, hypertension, osteoporosis, opportunistic infections **High-Yield:** Early addition of a steroid-sparing agent (azathioprine or mycophenolate) reduces cumulative corticosteroid exposure and long-term toxicity. 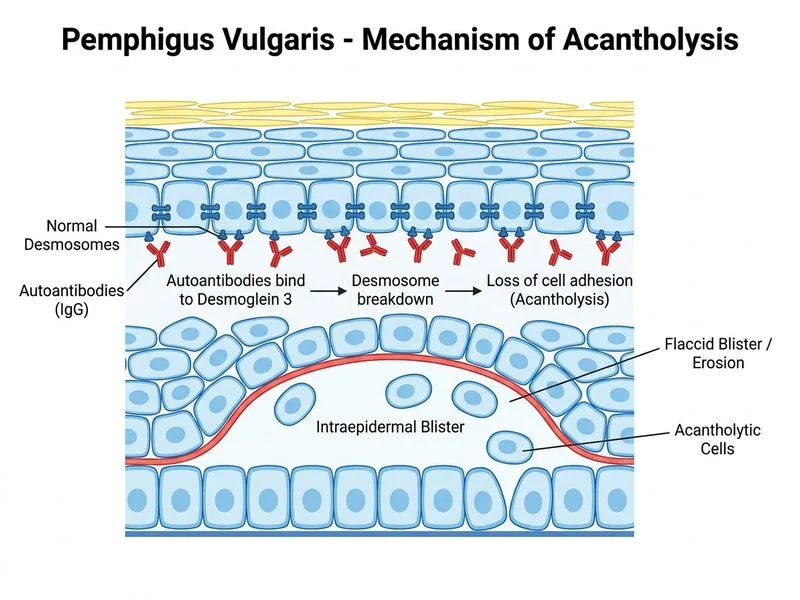
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.