## X-Linked Inheritance and G6PD Expression in Females ### Understanding X-Inactivation (Lyonization) **Key Point:** G6PD deficiency is an X-linked recessive disorder. Heterozygous females undergo random X-inactivation, resulting in two populations of RBCs: some expressing normal G6PD (from the active normal X chromosome) and some expressing deficient G6PD (from the active mutant X chromosome). **High-Yield:** This mosaicism protects heterozygous females from severe, recurrent haemolytic episodes because approximately 50% of their RBCs retain normal G6PD activity and can generate adequate NADPH to resist oxidative stress. ### Comparison: Heterozygous Female vs. Hemizygous Male | Feature | Heterozygous Female | Hemizygous Male | |---------|-------------------|------------------| | **G6PD-expressing RBCs** | ~50% (from active normal X) | 100% (single X chromosome) | | **G6PD-deficient RBCs** | ~50% (from active mutant X) | 100% (single X chromosome) | | **Average G6PD activity** | ~50% of normal | <10% of normal (depending on mutation) | | **Haemolytic episodes** | Milder, less frequent, triggered only by severe oxidative stress | Severe, recurrent, triggered by mild oxidative stress | | **Baseline haemolysis** | Usually absent or minimal | May be present even at baseline | | **Response to fava beans** | Mild to moderate haemolysis | Acute, life-threatening haemolysis | **Clinical Pearl:** The severity of G6PD deficiency in heterozygous females depends on the **skew of X-inactivation**. If inactivation is heavily skewed toward the normal X chromosome, the female may be clinically asymptomatic. If skewed toward the mutant X, she may have symptoms approaching those of hemizygous males. ### Molecular Basis of X-Inactivation ```mermaid flowchart TD A[Heterozygous Female<br/>X^N X^m]:::outcome --> B[Early Development<br/>Random X-inactivation]:::action B --> C[Cell Population 1<br/>X^N active, X^m inactivated]:::outcome B --> D[Cell Population 2<br/>X^m active, X^N inactivated]:::outcome C --> E[Normal G6PD expression<br/>NADPH production intact]:::action D --> F[Deficient G6PD expression<br/>NADPH production impaired]:::action E --> G[RBCs resistant to oxidative stress]:::outcome F --> H[RBCs susceptible to oxidative stress]:::outcome G --> I[Overall RBC population<br/>~50% protected]:::action H --> I I --> J[Milder haemolytic episodes<br/>than hemizygous males]:::outcome ``` **Mnemonic:** **LION** = **L**yonization (X-inactivation) **I**n **O**ne **N**ormal allele (heterozygous females). This protects them from severe haemolysis. ### Why This Patient's Course is Milder 1. **Mosaic RBC population:** Approximately 50% of her RBCs express normal G6PD and can generate sufficient NADPH to maintain GSH pools and defend against oxidative stress. 2. **Functional redundancy:** Even mild oxidative stress will preferentially affect the G6PD-deficient RBCs; the normal RBCs remain protected. 3. **Higher threshold for haemolysis:** A more severe oxidative insult is required to trigger haemolysis in heterozygous females compared to hemizygous males. 4. **Son's severe phenotype:** Her son, being hemizygous, has 100% of his RBCs expressing the mutant G6PD allele, making him vulnerable to haemolysis from even mild oxidative triggers. **Warning:** Do not confuse X-inactivation with gene dosage compensation. X-inactivation is the mechanism by which females achieve approximate dosage compensation with males for X-linked genes, but it results in cellular mosaicism, not uniform reduction of gene expression. 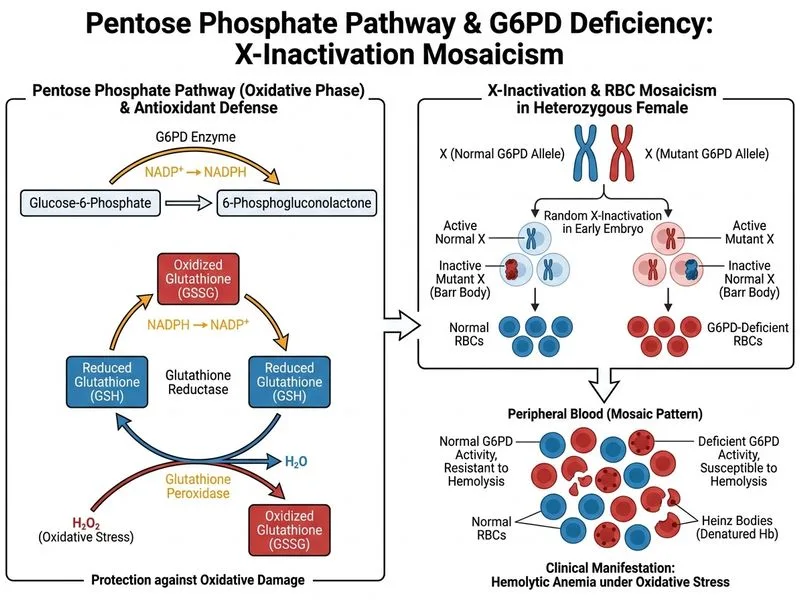
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.