## Tumor Marker Selection in Gastric Cancer ### Clinical Context This patient has biopsy-confirmed gastric adenocarcinoma with elevated CEA (8 ng/mL) and AFP (450 IU/mL). The question asks for the **most useful marker** for monitoring treatment response and detecting recurrence. ### Tumor Marker Profile in Gastric Cancer | Marker | Gastric Cancer Relevance | Sensitivity | Primary Use | |--------|--------------------------|-------------|-------------| | **CEA** | Most widely used | 30–50% | Monitoring treatment response, recurrence detection | | **CA 19-9** | Useful adjunct | 20–40% | Pancreatic/biliary > gastric; less validated for gastric monitoring | | **AFP** | Rare elevation (<5–10%) | Low | HCC, germ cell tumors; not a primary gastric marker | | **CA 72-4** | Elevated in gastric Ca | 50–70% | High specificity but not widely available | ### Why CEA is the Correct Answer **Key Point:** CEA is the **most widely validated and clinically used tumor marker for monitoring treatment response and detecting recurrence in gastric adenocarcinoma**. Per Harrison's Principles of Internal Medicine (21e, Ch. 91) and standard oncology guidelines (NCCN, ESMO): 1. **Established clinical utility**: CEA is routinely measured pre- and post-treatment in gastric cancer patients; serial CEA levels correlate with disease burden and treatment response. 2. **Already elevated in this patient**: CEA is 8 ng/mL (above normal <5), confirming it is a secreting marker in this tumor — making it directly applicable for monitoring. 3. **Guideline-endorsed**: NCCN and ESMO gastric cancer guidelines recommend CEA (along with CA 19-9) for post-treatment surveillance; CEA has stronger evidence for gastric cancer monitoring than CA 19-9 alone. 4. **CA 19-9 is primarily validated for pancreatic and biliary cancers**, not gastric adenocarcinoma specifically; its role in gastric cancer monitoring is secondary and less standardized. ### Why the Other Options Are Incorrect - **AFP (Option B):** Although elevated here (450 IU/mL), AFP elevation in gastric adenocarcinoma is rare and atypical; AFP is the primary marker for hepatocellular carcinoma and germ cell tumors. It is not routinely used to monitor gastric cancer treatment response. The elevated AFP in this case may reflect a rare AFP-producing gastric cancer variant or concurrent liver pathology, but CEA remains the standard monitoring marker. - **PSA (Option C):** Prostate-specific antigen is entirely irrelevant to gastric adenocarcinoma. - **CA 19-9 (Option D):** While CA 19-9 can be elevated in gastric cancer, it is primarily the marker of choice for **pancreatic and biliary** adenocarcinomas. CEA has stronger evidence and guideline support for gastric cancer monitoring specifically. **Clinical Pearl:** When a tumor marker is already elevated at diagnosis (as CEA is here at 8 ng/mL), that same marker becomes the most useful tool for monitoring — because you have a confirmed baseline to track. CEA is the standard-of-care marker for gastric adenocarcinoma surveillance per Harrison's and NCCN guidelines. ### High-Yield Summary - **Gastric adenocarcinoma monitoring** → **CEA** (primary), CA 19-9 (adjunct) - **Pancreatic/biliary cancer** → CA 19-9 (primary) - **HCC** → AFP - **Prostate cancer** → PSA [cite:Harrison 21e Ch. 91; NCCN Gastric Cancer Guidelines v2023] 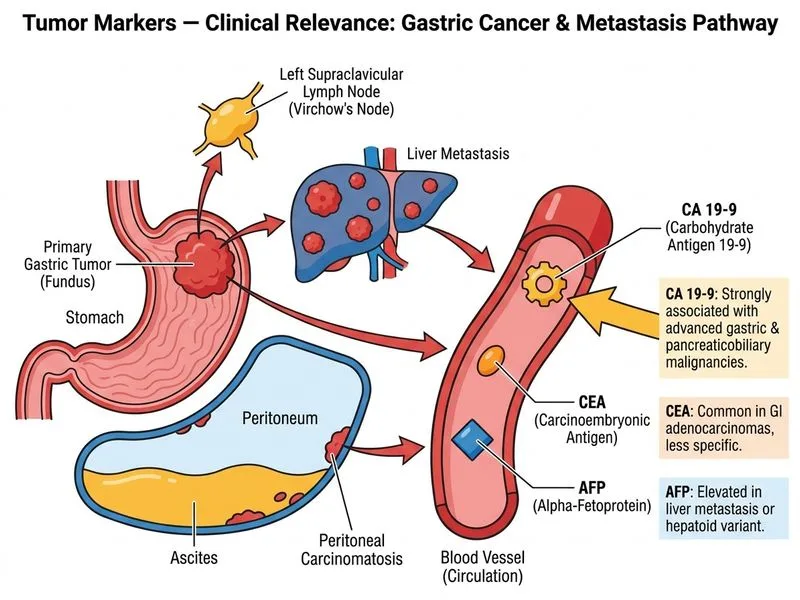
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.