## Confirming Urea Cycle Disorder in Acute Hyperammonemia ### Clinical Context In an adult with known or suspected urea cycle disorder presenting with acute encephalopathy, the challenge is to confirm that **hyperammonemia is the cause** and to **identify the specific enzyme defect** if not already known. **Key Point:** Plasma amino acid profiling is the most specific and direct investigation to confirm the urea cycle disorder because it reveals the characteristic amino acid pattern that identifies the exact enzymatic block. ### Why Plasma Amino Acid Profile is Superior | Investigation | What It Shows | Diagnostic Value | Timing | |---|---|---|---| | **Plasma amino acids** | Specific pattern of accumulation upstream of block | Identifies exact enzyme defect (CPS-I, OTC, ASS, ASL, ARG1) | 24–48 hrs | | **CSF ammonia/glutamine** | CNS ammonia metabolism | Confirms hyperammonemia effect on brain; not specific to urea cycle | Hours | | **Brain MRI spectroscopy** | ↑ Glutamine/glutamate, ↓ myo-inositol | Confirms hyperammonemia toxicity; non-specific | Hours–days | | **ABG + electrolytes** | Acid–base status, K^+^, Na^+^ | Assesses metabolic derangement; not diagnostic of urea cycle | ### Diagnostic Interpretation in OTC Deficiency **High-Yield:** In partial OTC deficiency (as in this case), plasma amino acid profiling shows: - **↓ Citrulline** (nearly absent or very low) — hallmark of OTC deficiency - **↑ Glutamine** (often >1000 µmol/L in acute crisis) - **↑ Alanine** - **↑ Aspartate** - **Normal or low arginine** - **Urine orotic acid** markedly elevated (secondary to shunting of carbamoyl phosphate to pyrimidine synthesis) This pattern is **pathognomonic** for OTC deficiency and distinguishes it from other causes of hyperammonemia (liver disease, portosystemic shunt, other urea cycle defects). ### Why Other Investigations Are Less Specific **Clinical Pearl:** CSF ammonia and glutamine reflect the **consequence** of hyperammonemia (glutamine accumulation in the CNS) but do not identify the **cause** (urea cycle defect). Brain MRI spectroscopy similarly shows the metabolic effect, not the defect itself. ### Diagnostic Algorithm ```mermaid flowchart TD A[Acute encephalopathy + ammonia 220]:::outcome --> B{Known urea cycle disorder?}:::decision B -->|Yes| C[Confirm with plasma amino acids]:::action B -->|No| D[Plasma amino acids to identify defect]:::action C --> E[Low citrulline = OTC confirmed]:::outcome D --> F[Pattern analysis]:::decision F -->|Low citrulline| G[OTC deficiency]:::outcome F -->|High citrulline + argininosuccinate| H[ASS deficiency]:::outcome F -->|High arginine| I[Arginase deficiency]:::outcome G --> J[Initiate ammonia-lowering therapy]:::action ``` **Tip:** In acute hyperammonemia, plasma amino acid profiling should be drawn **before** starting ammonia-lowering therapy (sodium benzoate, sodium phenylbutyrate, L-arginine) because treatment normalizes the amino acid pattern within hours, obscuring the diagnostic signature. 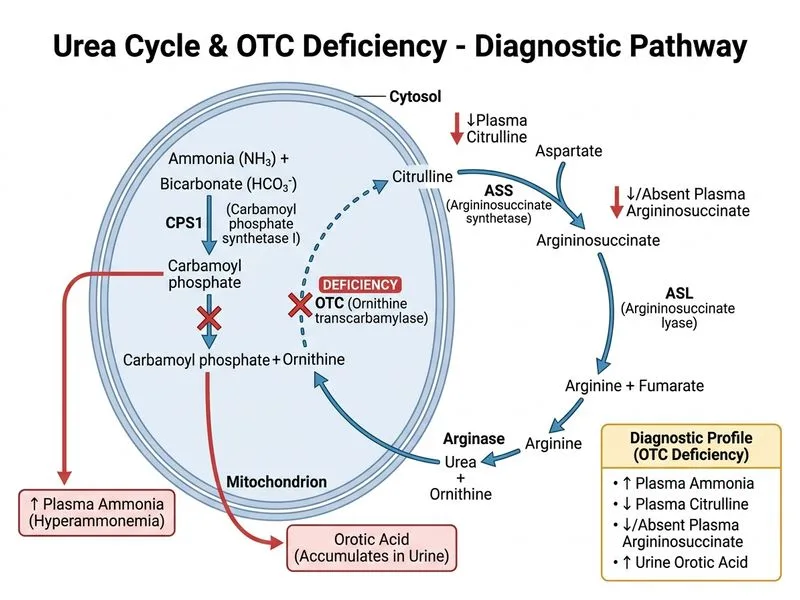
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.