Cardiovascular Physiology and Cardiac Cycle for NEET PG 2026
Master cardiac cycle, Wiggers diagram, ECG basis, heart sounds, preload-afterload, Starling's law, JVP waves, and CO measurement for NEET PG 2026 — high-yield MCQ traps.
Quick Answer
Cardiovascular physiology is a top-five NEET PG topic — expect 4 to 6 questions per paper across Physiology, Medicine, and Anaesthesia. Lock these:
- Cardiac cycle phases — 7 phases; systole 0.3 s plus diastole 0.5 s.
- Wiggers diagram — pressure, volume, ECG, heart sounds all on one timeline.
- Heart sounds — S1 (AV valve closure), S2 (semilunar valve closure), S3 (rapid filling), S4 (atrial contraction).
- JVP waves — a (atrial contraction), c (tricuspid bulge), v (atrial filling); x and y descents.
- Starling's law — stroke volume rises with end-diastolic volume.
- CO regulation — preload, afterload, contractility, heart rate.
- Fick principle — CO = VO2 / (CaO2 minus CvO2).
Cardiovascular physiology is the densest NEET PG block of pure concept-based questions because Wiggers, ECG vector analysis, and the JVP-PCWP coupling reward students who genuinely understand the cycle rather than just memorise tables. Examiners reach for it because each component — pressure-volume loops, JVP waves, heart sounds — has clean, exam-friendly clinical correlates.
This NEETPGAI deep dive walks through every phase of the cardiac cycle, the Wiggers diagram, ECG generation, heart sounds and murmurs, preload-afterload-contractility, Starling's law, neural and humoral regulation, and cardiac output measurement. Pair this guide with the arrhythmia management deep dive and the heart failure guide for a complete circulatory map.
The cardiac cycle — seven phases
At resting heart rate 75 beats per minute, each cycle is 0.8 seconds long — systole 0.3 s, diastole 0.5 s. Tachycardia shortens diastole disproportionately, which is why coronary perfusion (diastolic event) suffers most.
1. Atrial systole (last 0.1 s of diastole)
Atrial depolarisation (P wave) precedes contraction. Atria contribute 20 to 30 percent of ventricular end-diastolic volume — the "atrial kick", crucial in stiff ventricles (HFpEF, atrial fibrillation loses it).
Pressure events — small rise in atrial pressure (a wave on JVP) and ventricular pressure.
S4 if audible (always pathological in adults) — caused by atrial contraction against a stiff ventricle.
2. Isovolumetric ventricular contraction (0.05 s)
Ventricular depolarisation completes (QRS), ventricular pressure rises sharply, AV valves snap shut producing S1 (M1 then T1). All valves are closed; volume does not change while pressure climbs steeply.
Pressure events — c wave on JVP (tricuspid bulge into RA).
3. Rapid ejection (first 0.1 s of systole)
Ventricular pressure exceeds aortic/pulmonary pressure; semilunar valves open; about 70 percent of stroke volume ejected in this phase. Aortic pressure peaks.
ECG — between QRS and T.
4. Reduced ejection (last 0.15 s of systole)
Ventricular contraction wanes; ejection slows; ventricular and aortic pressures fall together. Repolarisation (T wave) starts.
5. Isovolumetric ventricular relaxation (0.08 s)
Ventricular pressure falls below aortic/pulmonary pressure; semilunar valves close producing S2 with physiological splitting (A2 then P2 on inspiration). AV valves still closed; volume unchanged. End-systolic volume reaches its minimum.
Pressure events — incisura (dicrotic notch) on aortic pressure trace from aortic valve closure; v wave peak on JVP.
6. Rapid filling (0.11 s)
Atrial pressure exceeds ventricular pressure; AV valves open with a y descent on JVP; blood rushes into the ventricle. If S3 is audible, it occurs here (physiological in young, pathological in volume overload or HFrEF in adults).
7. Slow filling (diastasis, 0.19 s at resting HR)
Ventricular filling slows as ventricle and atrium pressures equalise. This phase shortens most with tachycardia.
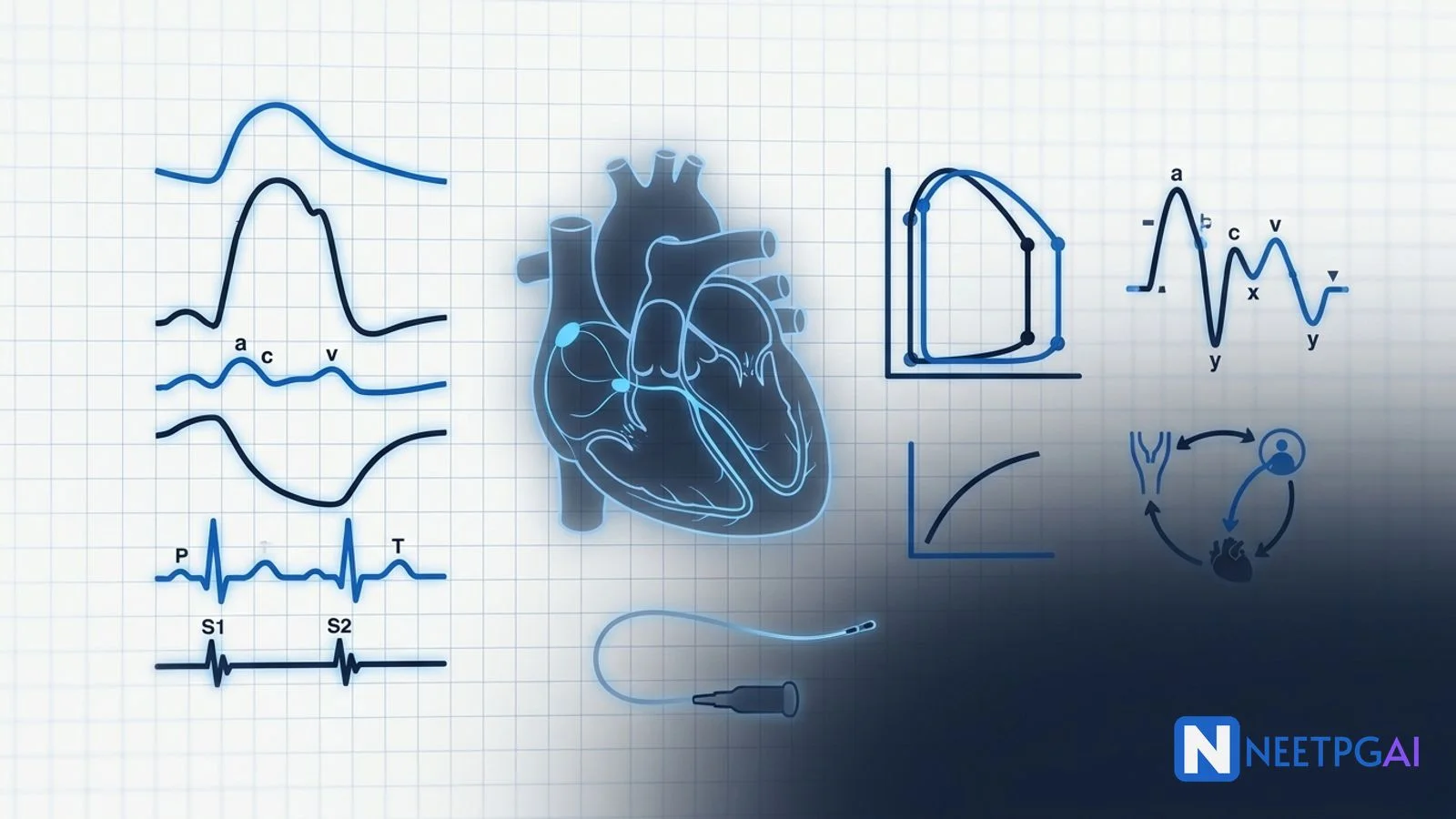
Wiggers diagram — read the whole cycle on one figure
The Wiggers diagram stacks (top to bottom) — aortic, ventricular, atrial pressures; ventricular volume; ECG; phonocardiogram (S1, S2, S3, S4); JVP waves. NEET PG questions often ask which event corresponds to which deflection. Anchor points:
- P wave triggers atrial contraction → a wave on JVP.
- QRS triggers ventricular contraction → S1 → c wave on JVP.
- End of QRS → aortic valve opens → start of rapid ejection.
- T wave end → aortic valve closes → S2 → dicrotic notch → v wave peak.
- Mitral valve opens → S3 (if present) → y descent.
Pressure-volume loops
Plot ventricular pressure (Y axis) against ventricular volume (X axis). A complete cycle traces a counter-clockwise rectangular loop.
- Bottom right corner = end-diastolic volume and pressure (LVEDV, LVEDP).
- Right vertical limb = isovolumetric contraction (volume constant, pressure rises).
- Top horizontal limb = ejection (volume falls, pressure peaks and declines).
- Top left corner = end-systolic volume and pressure (LVESV).
- Left vertical limb = isovolumetric relaxation (volume constant, pressure falls).
- Bottom horizontal limb = filling (volume rises along the end-diastolic pressure-volume curve).
Stroke volume = LVEDV minus LVESV (width of the loop).
Ejection fraction = SV / LVEDV (normal 55 to 70 percent).
Area within the loop = external stroke work.
End-systolic pressure-volume relationship (ESPVR) = contractility (steeper slope means stronger contraction).
End-diastolic pressure-volume relationship (EDPVR) = compliance (steeper means stiffer).
Loop changes
- Increased preload (volume loading) — wider loop, shifts right.
- Increased afterload — taller loop, narrower width, lower SV.
- Increased contractility — narrower loop with higher peak pressure, smaller LVESV, higher SV.
- HFrEF (systolic failure) — loop shifts right (dilated), reduced height, reduced width.
- HFpEF (diastolic dysfunction) — narrow stiff loop with high LVEDP and small LVEDV.
ECG basis — depolarisation and vectors
The ECG records the sum of all cardiac electrical activity from skin electrodes.
- P wave — atrial depolarisation (right then left atrium). Duration under 0.12 s.
- PR interval — atrial depolarisation plus AV nodal delay. Normal 0.12 to 0.20 s. Longer than 0.20 = first-degree AV block.
- QRS complex — ventricular depolarisation. Duration under 0.10 s; wider implies bundle-branch block or ventricular origin.
- ST segment — early ventricular repolarisation; isoelectric; elevation suggests ischaemia/STEMI, depression suggests subendocardial ischaemia.
- T wave — ventricular repolarisation; same direction as QRS in most leads.
- QT interval — total ventricular depolarisation plus repolarisation. Corrected QT (QTc, Bazett formula) less than 440 ms in men, less than 460 ms in women.
- U wave — late repolarisation; prominent in hypokalaemia.
Conduction system
SA node (right atrium, rate 60-100/min, sympathetic and parasympathetic innervation) → internodal pathways → AV node (rate 40-60/min, delay 0.1 s) → bundle of His → right and left bundle branches → left anterior and posterior fascicles → Purkinje fibres (rate 20-40/min) → ventricular myocardium.
The intrinsic pacemaker hierarchy explains escape rhythms — junctional 40-60/min if SA fails, ventricular 20-40/min if both fail.
Vector analysis
Mean QRS axis normally between -30 and +90 degrees in adults. Left axis deviation (-30 to -90) — LBBB, LVH, left anterior hemiblock, inferior MI. Right axis deviation (+90 to +180) — RVH, pulmonary embolism, COPD, left posterior hemiblock, lateral wall MI, dextrocardia.
Heart sounds and murmurs
Normal sounds
-
S1 (mitral and tricuspid closure) — start of systole; loudest at apex (M1) and lower left sternal border (T1). Loud S1 in mitral stenosis (stiff valve snaps shut), short PR. Soft S1 in mitral regurgitation (incomplete closure), first-degree heart block.
-
S2 (aortic and pulmonary closure) — end of systole. Normal physiological splitting on inspiration (A2 then P2 — increased venous return delays right ventricular emptying and P2). Wide fixed splitting in ASD. Paradoxical splitting in LBBB and aortic stenosis (P2 precedes A2). Loud P2 in pulmonary hypertension.
-
S3 — early diastolic; ventricular gallop; rapid filling of a dilated/non-compliant ventricle. Physiological in children, young adults, pregnancy. Pathological after age 40 — suggests HFrEF, volume overload, mitral regurgitation. Best heard at apex with bell.
-
S4 — late diastolic; atrial gallop; atrial contraction against stiff ventricle. Always pathological in adults — LVH, HFpEF, hypertensive heart disease, hypertrophic cardiomyopathy, AS, ischaemia. Absent in atrial fibrillation.
Murmurs by phase
| Murmur | Phase | Classic finding |
|---|---|---|
| Aortic stenosis | Mid-systolic ejection | Crescendo-decrescendo, radiates to carotids; pulsus parvus et tardus |
| Pulmonary stenosis | Mid-systolic | Left upper sternal border |
| Mitral regurgitation | Pansystolic | Apex, radiates to axilla |
| Tricuspid regurgitation | Pansystolic | Left lower sternal border, louder on inspiration (Carvallo sign) |
| Ventricular septal defect | Pansystolic | Left lower sternal border |
| Aortic regurgitation | Early diastolic | Left sternal border, leaning forward, expiration; collapsing pulse |
| Mitral stenosis | Mid-late diastolic | Apex, left lateral position, expiration; opening snap; loud S1 |
| Patent ductus arteriosus | Continuous | Machinery murmur, left infraclavicular |
Preload, afterload, contractility
Preload = end-diastolic ventricular volume (or wall stress at end-diastole). Approximated clinically by LVEDP, which itself is approximated by PCWP (pulmonary capillary wedge pressure). Increased by volume loading, venous return, supine posture; reduced by diuretics, venodilators (nitrates), haemorrhage.
Afterload = wall stress during ejection (Laplace law: T = P × r / 2h). Approximated clinically by systemic vascular resistance and aortic pressure. Increased by hypertension, aortic stenosis, vasoconstrictors; reduced by vasodilators (ACEi, hydralazine, nitroprusside).
Contractility (inotropy) = strength of contraction at a given preload and afterload. Increased by sympathetic stimulation (beta-1), circulating catecholamines, digoxin, dobutamine, milrinone, calcium; reduced by beta-blockers, calcium channel blockers, acidosis, ischaemia, hypoxia.
Frank-Starling's law
Within physiological limits, stroke volume rises with end-diastolic volume. Mechanism: optimal actin-myosin filament overlap at sarcomere length 2.2 micrometres maximises cross-bridge formation; longer sarcomeres also increase calcium sensitivity of troponin C.
Clinical relevance: explains how the right and left ventricles match outputs without external coordination, why volume expansion improves stroke volume in fluid-responsive shock, and why ventricular over-distension (HFrEF) drops onto the descending limb of the curve.
Cardiovascular regulation
Neural
- Baroreceptor reflex — carotid sinus and aortic arch stretch receptors → glossopharyngeal/vagus → nucleus tractus solitarius → modulates sympathetic and parasympathetic outflow. Fast (seconds). Reset after chronic hypertension.
- Chemoreflex — carotid and aortic bodies sense hypoxia, hypercapnia, acidosis → increase ventilation and sympathetic outflow.
Humoral
- RAAS — renin (juxtaglomerular cells respond to low renal perfusion, low Na delivery, beta-1 stimulation) → angiotensin II (vasoconstriction, aldosterone release, ADH release, thirst, cardiac and vascular remodelling) → aldosterone (Na retention, K excretion).
- ANP and BNP — released from atria/ventricles by stretch; cause natriuresis, vasodilation, suppression of renin and aldosterone. BNP is the biomarker of HF.
- ADH (vasopressin) — water retention via V2 collecting-duct receptors; vasoconstriction via V1 receptors.
Cardiac output measurement
Fick principle
CO = VO2 / (CaO2 minus CvO2)
Requires VO2 (250 mL/min at rest), arterial O2 content, and mixed-venous O2 content from pulmonary artery sample. Most accurate; impractical for routine use.
Thermodilution
Cold saline injected into the right atrium via Swan-Ganz catheter; temperature change measured downstream in the pulmonary artery. Modified Stewart-Hamilton equation calculates CO. Used in ICU.
Echocardiography
CO = SV × HR, where SV = LVOT area × LVOT velocity-time integral (VTI). Non-invasive.
Pulse contour analysis
Beat-to-beat CO from arterial line waveform (FloTrac, PiCCO).
NEET PG MCQ traps
- S2 splitting on inspiration — physiological; due to delayed P2 from increased venous return.
- Fixed splitting of S2 — atrial septal defect.
- Paradoxical splitting of S2 — LBBB, aortic stenosis (delayed A2).
- S3 in adults — HFrEF, volume overload, MR; physiological in young/pregnant.
- S4 — always pathological in adults; LVH, HFpEF, AS, HCM; absent in AF.
- Loud S1 — mitral stenosis, short PR, tachycardia.
- Pulsus parvus et tardus — aortic stenosis.
- Collapsing (water-hammer) pulse — aortic regurgitation.
- Pulsus paradoxus — cardiac tamponade, severe asthma; greater than 10 mmHg fall in SBP on inspiration.
- Pulsus alternans — severe LV dysfunction.
- Cannon a waves on JVP — third-degree AV block, ventricular tachycardia (atrial contraction against closed tricuspid).
- Large v waves on JVP — tricuspid regurgitation.
- PCWP approximates LA pressure — and LVEDP if no mitral disease.
- Aortic incisura (dicrotic notch) — aortic valve closure.
- Carvallo sign — inspiratory accentuation of right-sided murmurs (TR).
Recent updates and Indian context
- NMC Competency-Based UG Curriculum 2024 — emphasises mechanism-based physiology questions on cardiac cycle, JVP, heart sounds, and ECG-pressure correlation.
- NEXT (foundation phase) — physiology questions integrate with medicine clinical reasoning; expect Wiggers-diagram interpretation paired with valvular-disease vignettes.
- Indian context — rheumatic heart disease remains the commonest cause of mitral stenosis in NEET PG vignettes; congenital heart disease (VSD, ASD, PDA, tetralogy) features in paediatric stems given India's high birth rate.
- PCWP and Swan-Ganz — declining in ICU practice (ESCAPE, PAC-Man); echocardiography preferred. NEET PG still tests the principle.
Frequently asked questions
What are the phases of the cardiac cycle?
The cardiac cycle has 7 phases — atrial systole, isovolumetric ventricular contraction, rapid ejection, reduced ejection, isovolumetric ventricular relaxation, rapid filling, and slow (diastasis) filling. Systole comprises isovolumetric contraction plus ejection; diastole comprises isovolumetric relaxation plus filling plus atrial contraction. At rest, systole lasts about 0.3 s and diastole 0.5 s in a 0.8 s cycle (heart rate 75/min).
What causes the first and second heart sounds?
S1 is produced by closure of the mitral and tricuspid valves at the onset of ventricular systole — the M1 (mitral) component slightly precedes T1 (tricuspid). S2 is produced by closure of the aortic and pulmonary valves at the end of ventricular systole — A2 normally precedes P2, with physiological splitting on inspiration due to delayed P2 from increased venous return.
What do the a, c, and v waves of the JVP represent?
The a wave is right atrial contraction (occurs just before S1). The c wave is bulging of the closed tricuspid valve into the right atrium during isovolumetric ventricular contraction (occurs with S1). The v wave is passive right atrial filling against the closed tricuspid valve during ventricular systole (occurs around S2). The x descent follows the c wave (atrial relaxation); the y descent follows the v wave (tricuspid opening, rapid filling).
What is the Frank-Starling law of the heart?
Frank-Starling's law states that within physiological limits, the stroke volume of the heart increases as the end-diastolic volume (preload) increases. The mechanism is optimal overlap of actin and myosin filaments at longer sarcomere lengths (2.2 micrometres optimal), increasing the number of cross-bridges formed. This is intrinsic to the myocardium and operates independently of neurohormonal control. It enables the left and right ventricles to maintain matched outputs.
What is the Fick principle for cardiac output measurement?
The Fick principle states that cardiac output equals oxygen consumption (VO2) divided by the arteriovenous oxygen content difference (CaO2 minus CvO2). A patient with VO2 of 250 mL/min and an arteriovenous O2 difference of 50 mL/L has CO of 5 L/min. Mixed venous samples must come from the pulmonary artery (not the right atrium) for accuracy. Thermodilution via Swan-Ganz catheter is the modern alternative.
This content is for educational purposes for NEET PG exam preparation. It is not a substitute for professional medical advice, diagnosis, or treatment. Clinical information has been reviewed by qualified medical professionals.
Written by: NEETPGAI Editorial Team Reviewed by: Pending SME Review Last reviewed: May 2026
Ready to put this into practice?
Start practicing NEET PG MCQs with AI-powered explanations.
Start Free PracticeYour Next Step
Related Study Guides
Anxiety Disorders, OCD & PTSD for NEET PG 2026: Diagnosis, SSRI, CBT
Master anxiety, OCD, and PTSD for NEET PG 2026 — DSM-5 criteria, Y-BOCS, SSRI first-line, exposure-response prevention, ECT indications, India NMHS data.
Acute Appendicitis Management for NEET PG 2026: Diagnosis & Surgery
Master acute appendicitis for NEET PG 2026 — pathophysiology, Alvarado score, imaging, laparoscopic appendicectomy, antibiotic-only therapy, complications, MCQ traps.
Antihypertensive Drugs Pharmacology for NEET PG 2026: Classes, DOC, ADRs
Master antihypertensive pharmacology for NEET PG 2026 — ACEi, ARBs, CCBs, beta-blockers, diuretics, drug-of-choice, ADRs, and hypertensive emergency drugs.
Join our NEET PG community
Daily MCQs, study tips, and topper strategies on Telegram.
Join on Telegram →