## Distinguishing ESBL from AmpC β-Lactamase **Key Point:** The critical discriminator between ESBL and AmpC producers is the **inhibition by clavulanic acid**, a β-lactamase inhibitor. ### Mechanism of Inhibition ESBL enzymes (TEM, SHV, CTX-M variants) are serine β-lactamases that are irreversibly inhibited by clavulanic acid, a mechanism-based inhibitor. When clavulanic acid is added to the antibiotic susceptibility test: - ESBL-resistant organisms regain susceptibility to cephalosporins - The resistance pattern "reverts" in the presence of the inhibitor AmpC β-lactamases (chromosomal or plasmid-mediated) are NOT inhibited by clavulanic acid because they use a different catalytic mechanism (zinc-dependent or serine-based but structurally distinct). ### Comparative Features | Feature | ESBL | AmpC β-Lactamase | |---------|------|------------------| | **Clavulanic acid inhibition** | ✓ YES (diagnostic) | ✗ NO | | **Cefepime susceptibility** | Usually susceptible | Resistant | | **Carbapenem susceptibility** | Susceptible | Susceptible (unless additional mechanisms) | | **Ceftazidime** | Resistant | Resistant | | **Enzyme type** | Serine β-lactamase | Serine or metallo-β-lactamase | **High-Yield:** The **clavulanic acid inhibition test** is the gold standard for confirming ESBL production. A ≥3 mm increase in zone diameter around a cephalosporin disk when clavulanic acid is added confirms ESBL. **Clinical Pearl:** In clinical practice, ESBL producers typically remain susceptible to carbapenems and cefepime, making these the agents of choice for serious infections. AmpC producers also respond to carbapenems but NOT to cefepime. ### Why This Matters The clavulanic acid inhibition pattern directly reflects the biochemical nature of the enzyme and guides empiric therapy selection in resource-limited settings where molecular confirmation is unavailable. [cite:Harrison 21e Ch 139] 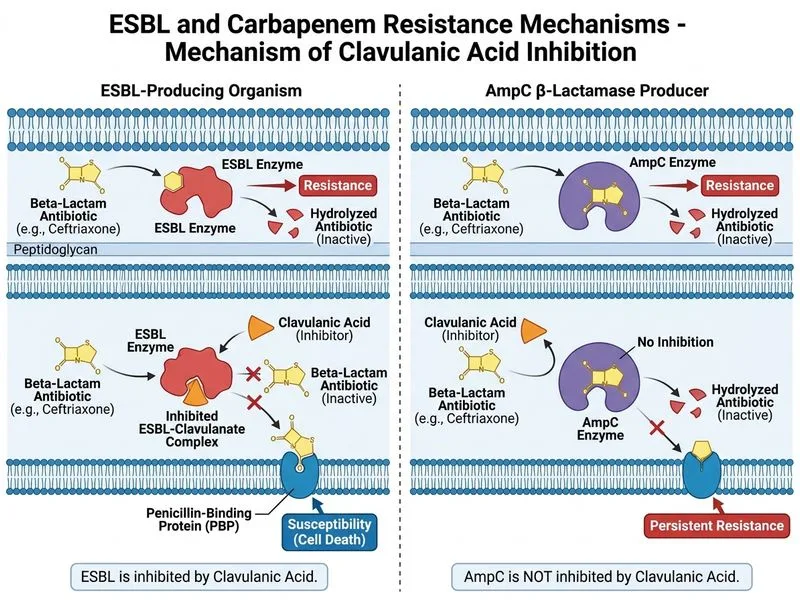
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.