## Biochemical Mechanism of Ketogenesis in DKA ### Normal Ketone Body Metabolism Ketone bodies (acetoacetate, β-hydroxybutyrate, and acetone) are produced in hepatic mitochondria from acetyl-CoA derived primarily from fatty acid β-oxidation. ### Why Ketogenesis Accelerates in Insulin Deficiency **Key Point:** In the absence of insulin, lipolysis is unopposed, releasing massive amounts of free fatty acids from adipose tissue. These undergo β-oxidation in hepatic mitochondria, generating excess acetyl-CoA. **High-Yield:** The rate-limiting step in ketogenesis is the availability of acetyl-CoA substrate. When acetyl-CoA production from fatty acid oxidation exceeds the oxidative capacity of the TCA cycle (which requires oxaloacetate, itself depleted by gluconeogenesis), acetyl-CoA is shunted into ketone body synthesis via the thiophorase pathway: 1. Acetyl-CoA + Acetyl-CoA → Acetoacetyl-CoA (via thiophorase) 2. Acetoacetyl-CoA + Acetyl-CoA → HMG-CoA (via HMG-CoA synthase) 3. HMG-CoA → Acetoacetate + Acetyl-CoA (via HMG-CoA lyase) 4. Acetoacetate ⇌ β-hydroxybutyrate (via β-hydroxybutyrate dehydrogenase) **Clinical Pearl:** In DKA, β-hydroxybutyrate predominates over acetoacetate (ratio ~3:1), making serum ketone measurement by nitroprusside (which detects acetoacetate) potentially underestimate true ketone burden. β-hydroxybutyrate assay is more accurate. ### Why This Patient Has Severe Ketosis - **Insulin deficiency** → unopposed lipolysis - **Glucagon excess** → further stimulates fatty acid mobilization and ketogenesis - **Depleted oxaloacetate** (diverted to gluconeogenesis) → TCA cycle cannot oxidize all incoming acetyl-CoA - **Result:** Massive acetyl-CoA accumulation → maximal ketone production ### Mnemonic: LACK - **L**ipolysis unopposed - **A**cetyl-CoA excess - **C**arbohydrate oxidation blocked (TCA cycle limited) - **K**etone production maximal 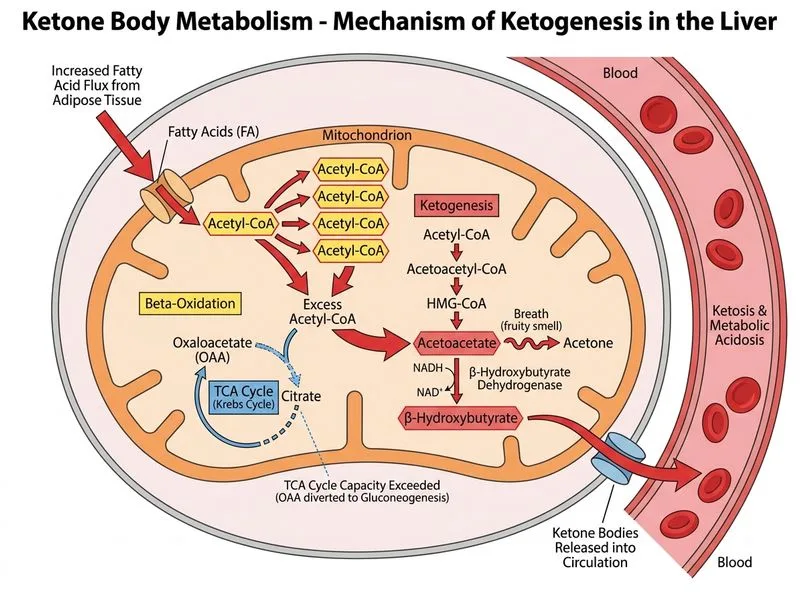
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.