## TNM Staging Analysis ### Tumour (T) Component - **6 cm left upper lobe mass with left main bronchus involvement** = **T4** - T4 is defined as: tumour with invasion of mediastinum, heart, great vessels, trachea, oesophagus, vertebral body, or carina; or separate tumour nodules in a different ipsilateral lobe. - Main bronchus involvement (within 2 cm of carina) qualifies as T4. ### Node (N) Component - **Ipsilateral hilar lymphadenopathy** = N1 - **Contralateral mediastinal lymph node enlargement (station 4R)** = **N3** - N3 is defined as: ipsilateral supraclavicular/scalene nodes OR contralateral mediastinal/hilar nodes OR contralateral supraclavicular nodes. - Contralateral mediastinal involvement (station 4R) is explicitly N3. ### Metastasis (M) Component - **Pleural effusion with negative cytology** = **M0** (not M1a) - **Key Point:** Malignant pleural effusion is classified as M1a in the 8th edition TNM staging (2017). However, a **negative pleural fluid cytology** in the context of a pleural effusion associated with a primary lung cancer is still considered a **pleural effusion related to the primary tumour** and is classified as **T4** (if it causes airway obstruction or involves adjacent structures) or as a **separate consideration**. In this case, the pleural effusion is a consequence of the large mass and mediastinal involvement, NOT a distant metastasis. Therefore, M0 is appropriate. - No distant organ metastases mentioned = M0. ### Overall Stage **T4N3M0 = Stage IIIB** [cite: AJCC 8th edition TNM staging for lung cancer] ## Treatment for Stage IIIB NSCLC **High-Yield:** Stage IIIB NSCLC (T4N3M0 or any T/N with N3) is **unresectable** and requires **concurrent chemoradiotherapy** as the standard of care for fit patients. ### Rationale for Concurrent Chemoradiotherapy 1. **Unresectable disease:** Contralateral mediastinal lymph node involvement (N3) and T4 status (main bronchus involvement) make this patient a non-surgical candidate. 2. **Curative intent:** Concurrent chemoradiotherapy (not sequential) offers superior survival compared to sequential or radiotherapy alone (RTOG 9410 trial). 3. **Chemotherapy regimen:** Cisplatin-based doublet (e.g., cisplatin + etoposide or cisplatin + pemetrexed) concurrent with thoracic radiotherapy (60 Gy in 30 fractions). 4. **Squamous cell histology:** Cisplatin + etoposide is preferred for squamous cell carcinoma. **Clinical Pearl:** Contralateral mediastinal lymph node involvement (N3) is a hallmark of unresectable disease and mandates concurrent chemoradiotherapy. Surgical resection is not indicated even with neoadjuvant chemotherapy. ## Why Other Options Are Incorrect **Neoadjuvant chemotherapy followed by surgery:** Contralateral mediastinal lymph node involvement (N3) makes this patient unresectable. Neoadjuvant chemotherapy does not convert N3 disease to resectable status in most cases. Surgery is contraindicated. **Palliative chemotherapy (Stage IV):** This is stage IIIB, not stage IV. The pleural effusion with negative cytology is not classified as M1a (malignant pleural effusion). Curative intent with concurrent chemoradiotherapy is appropriate. **Adjuvant radiotherapy after surgery:** Surgery is not indicated for T4N3M0 disease. Adjuvant radiotherapy is given after surgical resection, not as primary treatment for unresectable stage IIIB NSCLC. 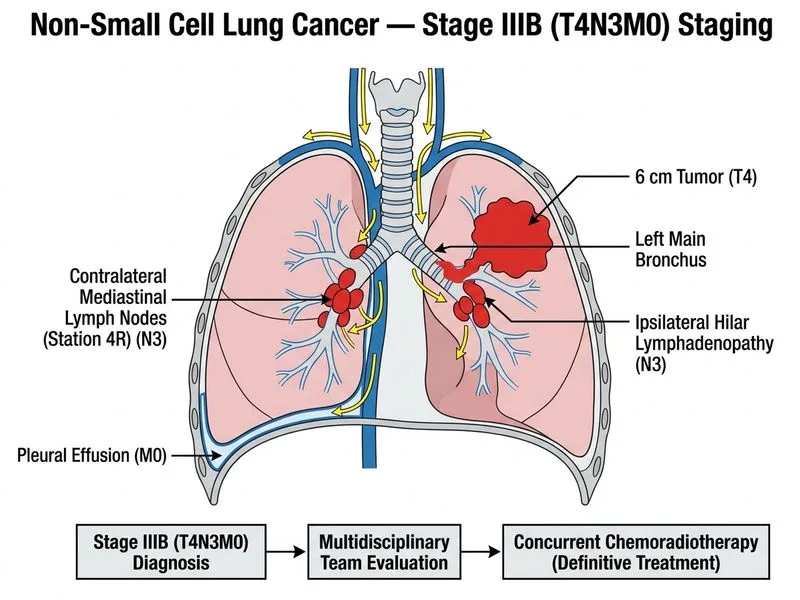
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.