## Investigation of Choice for Hilar Mass with Pleural Effusion ### Clinical Context A hilar lung mass with pleural effusion raises concern for advanced non-small cell lung cancer (NSCLC) with pleural involvement. The negative initial thoracentesis cytology does not exclude malignancy; pleural cytology sensitivity is only 40–60% on first tap and improves to ~80% with repeat sampling or alternative techniques. However, the primary goal here is to establish tissue diagnosis of the lung mass itself and assess mediastinal lymph node involvement. ### Why EBUS-TBNA is Optimal **Key Point:** EBUS-TBNA is the gold standard for sampling hilar and mediastinal lymph nodes and is superior to transbronchial biopsy for central masses. It provides both diagnosis and staging in a single procedure [cite:Harrison 21e Ch 87]. 1. **Direct Lymph Node Access**: EBUS allows real-time ultrasound visualization of hilar (station 10L/R) and mediastinal lymph nodes (stations 2–9), enabling precise needle placement. 2. **High Diagnostic Yield**: Sensitivity 85–95% for malignancy in enlarged lymph nodes; superior to blind transbronchial biopsy (50–70%). 3. **Staging Information**: Simultaneous assessment of N-stage (mediastinal involvement) guides treatment decisions (surgery vs. chemoradiotherapy). 4. **Pleural Involvement Assessment**: Biopsies of involved lymph nodes and adjacent tissue help determine resectability and stage. 5. **Minimally Invasive**: Performed via flexible bronchoscope; no general anaesthesia required; low morbidity. ### Comparison of Diagnostic Approaches | Investigation | Target | Yield for Hilar Mass | Staging Capability | Invasiveness | |---|---|---|---|---| | **EBUS-TBNA** | Hilar/mediastinal nodes | 85–95% | Excellent (N-staging) | Bronchoscopic, low risk | | **Repeat thoracentesis** | Pleural fluid | 40–80% (depends on volume, technique) | None for lung mass | Low risk but limited diagnostic value | | **Transoesophageal EUS** | Posterior mediastinal nodes (stations 5, 8, 9) | 80–90% for posterior nodes | Partial (limited to posterior stations) | Endoscopic, moderate risk | | **Mediastinoscopy** | Anterior/middle mediastinal nodes (stations 2, 3, 4) | 90–95% | Excellent but invasive | Surgical, general anaesthesia, higher morbidity | **Clinical Pearl:** EBUS-TBNA has largely replaced mediastinoscopy as the first-line invasive staging tool for suspected NSCLC because it is less invasive, has comparable diagnostic yield, and can be combined with transbronchial biopsy of the primary mass if accessible. ### Diagnostic Algorithm for Hilar Mass ```mermaid flowchart TD A[Hilar Mass on CXR/CT]:::outcome --> B{Endobronchial involvement?}:::decision B -->|Yes, visible at bronchoscopy| C[Transbronchial Biopsy]:::action B -->|No, or mass not visible| D[EBUS-TBNA of hilar/mediastinal nodes]:::action C --> E[Tissue Diagnosis + EBUS for N-staging]:::action D --> F[Tissue Diagnosis + N-staging]:::outcome F --> G{Pleural Effusion Present?}:::decision G -->|Yes, cytology negative| H[Consider Pleural Biopsy or Pleuroscopy if diagnosis unclear]:::action G -->|No| I[Proceed to Full Staging: PET-CT, Brain MRI]:::action H --> I I --> J[Treatment Planning]:::outcome ``` **High-Yield:** EBUS-TBNA is the preferred first-line invasive investigation for hilar/mediastinal masses because it provides both diagnosis and staging with minimal morbidity. Mediastinoscopy is reserved for cases where EBUS is non-diagnostic or when anterior mediastinal nodes need biopsy. 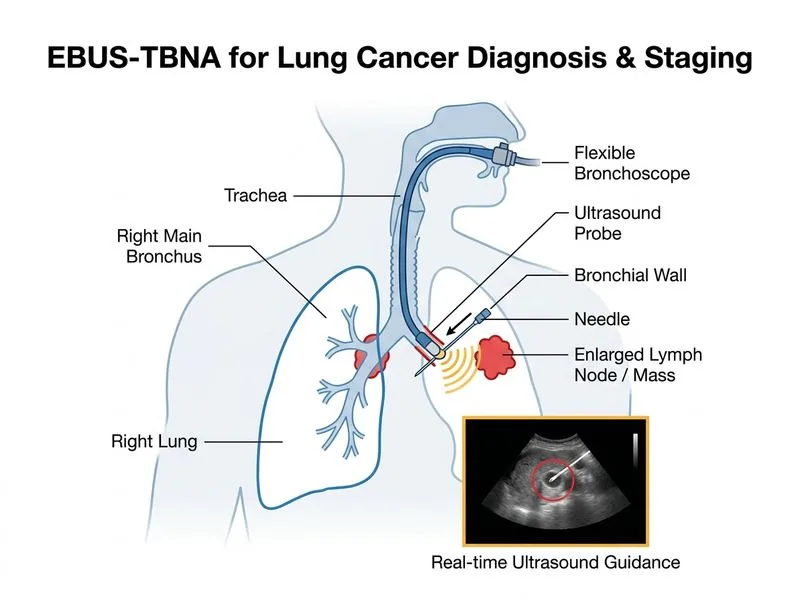
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.