## Clinical Diagnosis This patient has **secondary osteoporosis in the setting of chronic kidney disease (CKD) with secondary hyperparathyroidism (SHPT)** — a fundamentally different pathophysiology from primary postmenopausal osteoporosis. **Key Point:** The elevated PTH (280 pg/mL) with hypocalcemia (Ca 7.8 mg/dL), hyperphosphatemia (PO₄ 5.2 mg/dL), and low 25-OH vitamin D (12 ng/mL) indicates **CKD-mineral bone disorder (CKD-MBD)**, not simple osteoporosis. This requires metabolic correction before or concurrent with bisphosphonate therapy. ## Why Calcitriol + Phosphate Binder Is Correct **High-Yield:** In CKD-MBD with SHPT, the treatment hierarchy per **KDIGO 2017 CKD-MBD guidelines** is: 1. **Correct vitamin D deficiency** → Use calcitriol (active 1,25-dihydroxyvitamin D) because CKD kidneys have impaired 1α-hydroxylase activity and cannot efficiently convert 25-OH vitamin D to its active form. Nutritional vitamin D supplementation alone is insufficient. 2. **Control phosphate** → Phosphate binders (calcium-based or non-calcium-based) reduce serum phosphate, which in turn suppresses PTH secretion and reduces vascular calcification risk. 3. **Normalize PTH** → KDIGO 2017 targets PTH at **2–9× the upper limit of normal** in CKD G3b–G5 (approximately 130–585 pg/mL for this lab's reference range); this patient's PTH of 280 pg/mL is within the acceptable range but metabolic derangements (hypocalcemia, hyperphosphatemia) must still be corrected. 4. **Defer bisphosphonates** → Once metabolic parameters are optimized; bisphosphonates suppress bone turnover and may worsen **adynamic bone disease** if used prematurely in the setting of uncontrolled SHPT. **Clinical Pearl:** KDIGO 2017 suggests that in CKD G3a–G5 (eGFR 15–59 mL/min), bisphosphonates may be used with caution when eGFR is 30–45 mL/min (stage G3b), but **only after correcting metabolic abnormalities** (hypocalcemia, hyperphosphatemia, vitamin D deficiency). This patient's eGFR of 35 mL/min (CKD stage G3b) places him in a borderline zone — bisphosphonates are not absolutely contraindicated, but initiating them before metabolic correction is inappropriate and potentially harmful. ## Mechanism of CKD-MBD ```mermaid flowchart TD A[Chronic Kidney Disease eGFR < 45]:::outcome --> B[↓ 1α-hydroxylase activity]:::outcome B --> C[↓ Calcitriol production]:::outcome C --> D[↓ Serum calcium]:::outcome D --> E[↑ PTH secretion]:::outcome E --> F[Secondary Hyperparathyroidism]:::outcome C --> G[↓ Phosphate excretion]:::outcome G --> H[↑ Serum phosphate]:::outcome H --> E E --> I[High bone turnover + osteoporosis]:::outcome J[Treatment Strategy]:::action --> K[Calcitriol 0.25-0.5 μg BID]:::action J --> L[Phosphate binder]:::action J --> M[Calcium supplementation]:::action K --> N[Target PTH 2-9× ULN per KDIGO 2017]:::outcome L --> N M --> N N --> O{Metabolic parameters corrected?}:::decision O -->|Yes| P[Consider bisphosphonate if fracture risk remains high]:::action O -->|No| Q[Adjust calcitriol/binder dose]:::action ``` ## Why Not the Other Options | Option | Why Incorrect | |--------|---------------| | **Alendronate monotherapy (A)** | Initiating bisphosphonates without first correcting hypocalcemia, hyperphosphatemia, and vitamin D deficiency is inappropriate. Bisphosphonates can worsen hypocalcemia acutely and, in the setting of uncontrolled SHPT with high bone turnover, risk precipitating adynamic bone disease. Oral bisphosphonates also have reduced GI absorption and altered pharmacokinetics in CKD. | | **IV zoledronic acid (C)** | Zoledronic acid is nephrotoxic and is generally avoided when eGFR < 35 mL/min (this patient is at the threshold). More importantly, "aggressive vitamin D repletion" with cholecalciferol/ergocalciferol without phosphate control and calcitriol is insufficient in CKD. Metabolic correction must precede bisphosphonate use. | | **Denosumab monotherapy (B)** | Denosumab (RANKL inhibitor) suppresses bone turnover and carries a **particularly high risk of severe, prolonged hypocalcemia in CKD patients** because: (1) this patient is already hypocalcemic (Ca 7.8 mg/dL), and (2) CKD impairs the compensatory PTH-mediated calcium mobilization. Denosumab is not first-line in CKD-MBD and should only be used after metabolic stabilization with close calcium monitoring. | ## KDIGO 2017 CKD-MBD Management Targets (Stage G3b–G4) | Parameter | Target | |-----------|--------| | Serum calcium | Normal range (8.5–10.2 mg/dL) | | Serum phosphate | Normal range (2.5–4.5 mg/dL) | | PTH | 2–9× upper limit of normal | | 25-OH vitamin D | Replete if deficient (≥30 ng/mL) | **Mnemonic:** **CKD-MBD First = Calcitriol + Control phosphate + Correct PTH; Bisphosphonate Later (only after metabolic correction)** ## Fracture Risk in CKD Paradoxically, CKD patients have **high fracture risk despite elevated PTH and high bone turnover** due to: - Poor bone quality (marrow fibrosis, impaired mineralization) - Vascular calcification - Medication side effects (steroids, diuretics) - Falls from uremia, anemia, and neuropathy Therefore, metabolic correction takes priority over bisphosphonate therapy in this clinical scenario. *Reference: KDIGO 2017 Clinical Practice Guideline Update for the Diagnosis, Evaluation, Prevention, and Treatment of Chronic Kidney Disease–Mineral and Bone Disorder (CKD-MBD); Harrison's Principles of Internal Medicine, 21st ed., Chapter on Renal Osteodystrophy.* 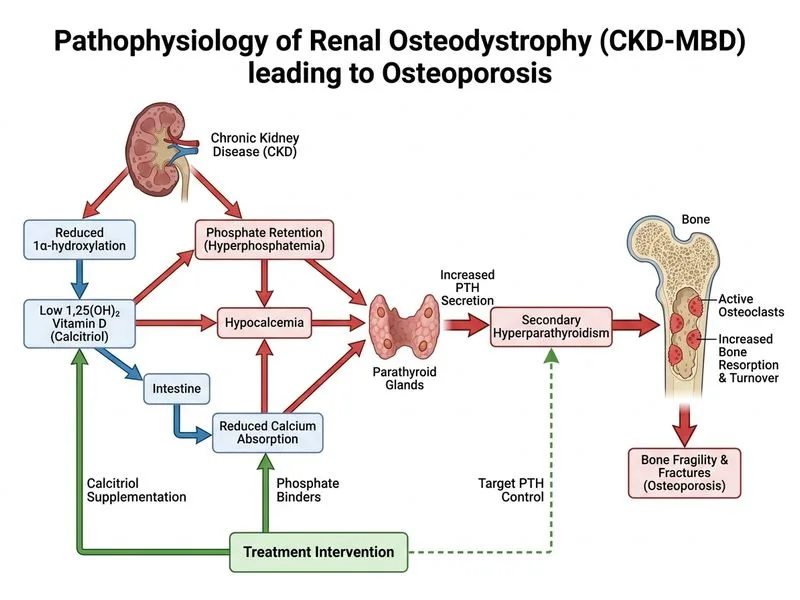
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.