## FIGO Staging Assessment **Key Point:** This patient has bilateral ovarian involvement with omental and peritoneal metastases but no distant organ involvement — this defines **FIGO Stage IIIC** (peritoneal metastases beyond pelvis and/or positive retroperitoneal lymph nodes). ### Staging Breakdown - **Tumor extent:** Bilateral ovaries (Stage III criterion) - **Distribution:** Omental nodules + peritoneal involvement (IIIC, not IIIA or IIIB) - **Ascites alone:** Does NOT upstage; nodules/deposits do - **No distant metastases:** Rules out Stage IV ## First-Line Chemotherapy for Advanced Ovarian Cancer **High-Yield:** For Stage IIIC-IV epithelial ovarian cancer (EOC), the standard of care is **Carboplatin + Paclitaxel (TC)** administered intravenously, typically for 6 cycles after optimal cytoreduction. ### Rationale for TC Regimen 1. **Platinum-based backbone:** Carboplatin is preferred over cisplatin in most patients (less nephrotoxicity, neurotoxicity) 2. **Taxane synergy:** Paclitaxel enhances platinum efficacy and is standard in advanced EOC 3. **Evidence base:** ICON3 and GOG-111 trials established TC as first-line for advanced disease 4. **High-grade serous histology:** Platinum-sensitive; responds optimally to TC ### Role of Bevacizumab **Clinical Pearl:** Bevacizumab (anti-VEGF) may be added to TC in newly diagnosed Stage IV or recurrent platinum-sensitive disease (GOG-218, ICON7 trials), but it is **not standard in Stage IIIC alone** unless there are additional risk factors or in the context of clinical trials. Primary recommendation remains **TC alone** for this patient. ### Why Not Other Regimens? - **CAP (Cisplatin + Doxorubicin + Cyclophosphamide):** Older regimen; TC superior in modern trials - **Carboplatin + Gemcitabine:** Reserved for platinum-resistant or second-line settings [cite:NCCN Ovarian Cancer Guidelines 2023; FIGO Staging 2014] ## Summary Table: EOC First-Line Chemotherapy by Stage | Stage | Primary Surgery | Chemotherapy | Notes | | --- | --- | --- | --- | | IA–IB (low-risk) | Staging laparotomy | Observation or TC | Age <40, Grade 1–2 may observe | | IA–IB (high-risk), IC–IIA | Optimal cytoreduction | Carboplatin + Paclitaxel | 6 cycles IV | | IIB–IV | Optimal cytoreduction ± NACT | Carboplatin + Paclitaxel ± Bevacizumab | Add bevacizumab in Stage IV or select IIIC | 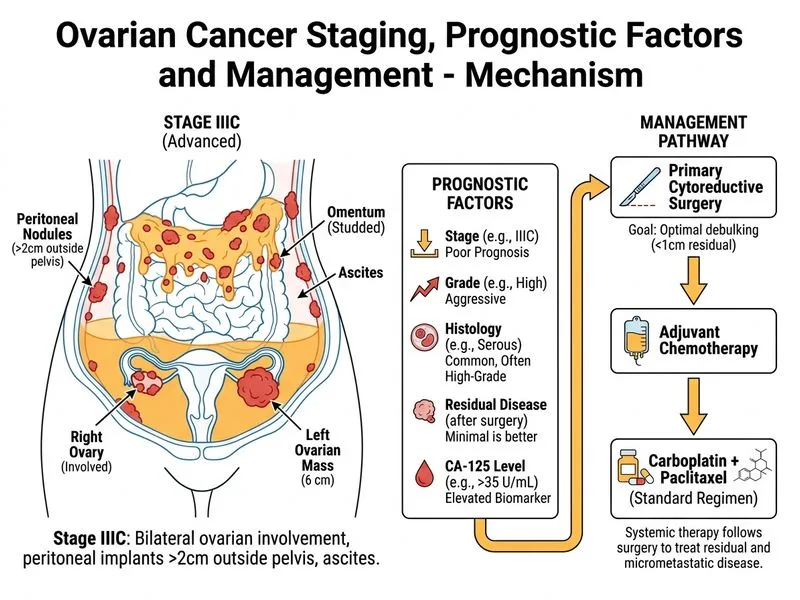
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.