## Investigation of Choice: Genomic Testing for BRCA1/BRCA2 Mutations and HRD Score ### Prognostic and Predictive Significance in Ovarian Cancer **Key Point:** HRD status (homologous recombination deficiency) is a critical prognostic and predictive biomarker in ovarian cancer that: - Predicts sensitivity to platinum-based chemotherapy - Determines eligibility for PARP inhibitor (olaparib, rucaparib, niraparib) maintenance therapy - Identifies patients with germline or somatic BRCA1/BRCA2 mutations - Guides long-term surveillance and family counseling ### HRD Testing Methods and Clinical Application **High-Yield:** HRD status is assessed via: 1. **BRCA1/BRCA2 mutation testing** — germline and somatic mutations 2. **HRD score** — composite genomic instability score (loss of heterozygosity, telomeric allelic imbalance, large-scale state transitions) measured by next-generation sequencing (NGS) or specialized platforms (e.g., Myriad myChoice®, Invitae) 3. **Combined assessment** — BRCA mutation status + HRD score determines PARP inhibitor eligibility ### PARP Inhibitor Eligibility and Maintenance Therapy | HRD Status | BRCA Status | PARP Inhibitor Eligibility | Mechanism | |---|---|---|---| | **HRD-positive** | BRCA1/2 mutated | Olaparib, rucaparib, niraparib | Synthetic lethality in HR-deficient cells | | **HRD-positive** | BRCA wild-type (sHRD) | Niraparib (preferred); rucaparib | Genomic instability compensates for BRCA loss | | **HRD-negative** | BRCA wild-type | Bevacizumab ± PARP (limited benefit) | No homologous recombination deficiency | **Clinical Pearl:** In this case (stage IIIC high-grade serous adenocarcinoma), HRD testing is performed on the surgical specimen to: - Confirm platinum sensitivity (expected in ~50% of HGS ovarian cancers) - Determine PARP inhibitor maintenance therapy after platinum-based chemotherapy - Identify BRCA carriers for genetic counseling and familial surveillance ### Comparison with Other Modalities | Investigation | Purpose | Limitation in This Context | |---|---|---| | **IHC for BRCA1/2** | Screening for BRCA loss (surrogate); not definitive | Indirect assessment; cannot quantify HRD score; poor concordance with mutation status | | **Genomic testing (HRD panel)** | Definitive BRCA mutation + HRD score | Gold standard; required for PARP inhibitor eligibility | | **Flow cytometry** | Prognostic assessment (ploidy, proliferation) | Outdated; does not guide targeted therapy; not relevant for HRD/PARP decisions | | **FISH for HER2** | Trastuzumab eligibility in HER2+ tumors | Not applicable; ovarian cancer is rarely HER2-driven; does not assess HRD | **Warning:** IHC for BRCA1 loss is a screening tool only and has poor positive predictive value for BRCA mutations. Definitive genomic testing is mandatory before PARP inhibitor prescription. ### Treatment Algorithm Context ```mermaid flowchart TD A[Stage IIIC High-Grade Serous Ovarian Cancer]:::outcome --> B[Cytoreductive Surgery + Tissue Sampling]:::action B --> C[Genomic Testing: BRCA + HRD Score]:::action C --> D{HRD-Positive?}:::decision D -->|Yes: BRCA mutated| E[Platinum-based chemotherapy + Olaparib maintenance]:::action D -->|Yes: BRCA WT, sHRD| F[Platinum-based chemotherapy + Niraparib maintenance]:::action D -->|No: HRD-negative| G[Platinum-based chemotherapy + Bevacizumab ± PARP]:::action E --> H[Genetic counseling; family screening]:::action ``` **High-Yield:** PARP inhibitor maintenance therapy (olaparib, rucaparib, niraparib) is now standard of care for HRD-positive ovarian cancer after platinum-based chemotherapy, improving progression-free survival and overall survival. [cite:NCCN Ovarian Cancer Guidelines 2023; ASCO Guideline on BRCA Testing and PARP Inhibitors in Ovarian Cancer] 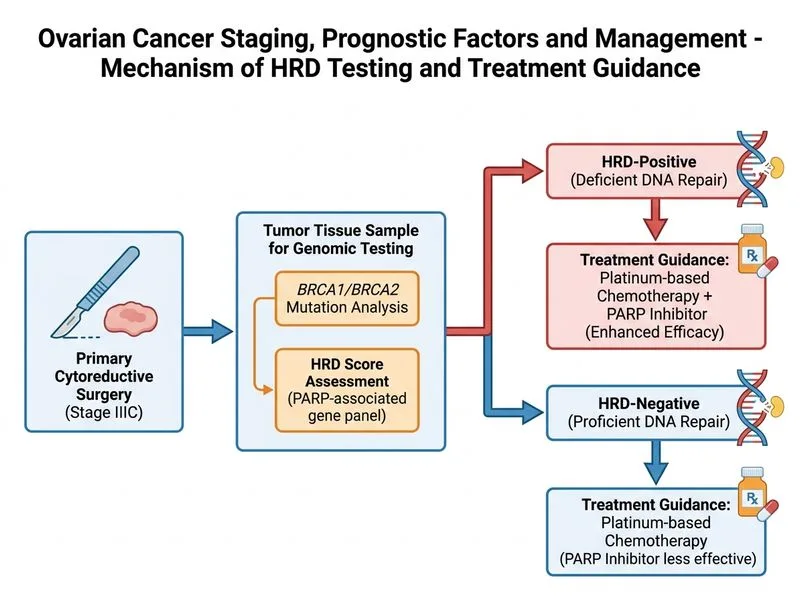
Sign up free to access AI-powered MCQ practice with detailed explanations and adaptive learning.