Ovarian Cysts and Tumors for NEET PG 2026: Diagnosis, Staging, Treatment
Master ovarian cysts and tumors for NEET PG 2026 — functional cysts, benign and malignant tumors, RMI, IOTA, FIGO 2014 staging, BRCA, chemotherapy, MCQ traps.
Quick Answer
Ovarian pathology is a NEET PG heavyweight across OBG and Pathology — expect 3 to 5 questions per paper. Lock these:
- Functional cysts — follicular (commonest), corpus luteum, theca lutein (associated with molar pregnancy and ovulation induction).
- Benign tumors — serous and mucinous cystadenoma, mature cystic teratoma (dermoid — commonest in young women).
- Malignant tumors — serous carcinoma (commonest epithelial), germ cell (dysgerminoma — young), sex cord stromal (granulosa cell — estrogenic).
- RMI greater than 200 — refer to gynaeoncology.
- FIGO 2014 staging — surgical staging; debulking plus chemotherapy.
- BRCA testing — all high-grade epithelial cancers regardless of family history.
- Meigs syndrome — benign fibroma plus ascites plus right pleural effusion.
Ovarian cancer is the second-commonest gynaecological malignancy in Indian women after cervical, with an age-standardised incidence of 6.7 per 100,000 (ICMR-NCRP 2023). It is also the deadliest because 70 percent of cases present at advanced stage owing to non-specific symptoms. NEET PG examiners reach for ovarian pathology because the differential diagnosis trees are clean, the staging is surgical, and the chemotherapy regimens are well-defined.
This NEETPGAI deep dive walks through every cyst category, every tumour subtype, risk stratification with RMI and IOTA Simple Rules, FIGO 2014 surgical staging, contemporary chemotherapy, BRCA-driven risk-reducing surgery, and India-specific late-presentation challenges. Pair this guide with the PCOS deep dive for the complete adnexal-pathology map.
Functional cysts
Functional cysts arise from the normal cyclical ovulatory process and represent the commonest "adnexal mass" encountered in reproductive-age women.
Follicular cyst
Pathology — failure of follicular rupture during ovulation; the unruptured follicle continues to grow. Lined by granulosa cells.
Size — 3 to 8 cm. Above 10 cm is rare.
Clinical — usually asymptomatic, occasionally menstrual irregularity, pelvic discomfort.
Management — observe; most resolve within 1 to 2 menstrual cycles. Repeat USG at 6 to 12 weeks. Surgery only for persistent cysts above 8 cm, suspicious features, or torsion.
Corpus luteum cyst
Pathology — failure of corpus luteum involution after ovulation. Lined by luteinised granulosa and theca cells; haemorrhagic content common.
Clinical — delayed menses, pelvic pain, sometimes acute pain from rupture (classic mimic of ectopic pregnancy — always beta-HCG). Halban triad: amenorrhoea, abdominal pain, adnexal mass with negative HCG.
Management — observe; haemodynamically stable rupture managed conservatively; emergency surgery if haemoperitoneum.
Theca lutein cysts
Pathology — bilateral, multilocular, large cysts caused by markedly elevated beta-HCG.
Associations — complete hydatidiform mole, choriocarcinoma, multiple gestation, ovarian hyperstimulation syndrome.
Management — resolve spontaneously after the underlying HCG-producing process is treated.
Benign tumors
Serous cystadenoma
Pathology — unilocular, thin-walled cyst lined by ciliated tubal-type epithelium; clear serous fluid. Bilateral in 15 to 25 percent.
Demographics — peak age 30 to 50 years.
Treatment — cystectomy in young women, oophorectomy in older or post-menopausal.
Mucinous cystadenoma
Pathology — multilocular, large (often above 15 cm — "ovarian giant"), lined by intestinal or endocervical-type mucin-producing epithelium. Rarely bilateral (5 percent).
Complication — rupture causing pseudomyxoma peritonei (mucinous ascites — often actually appendiceal in origin).
Mature cystic teratoma (dermoid)
Pathology — germ cell tumour containing tissues from all three germ layers (ectoderm dominant — hair, sebaceous material, teeth). The classic "Rokitansky protuberance" on USG.
Demographics — commonest ovarian tumour in young women (10 to 30 years).
Imaging — heterogeneous mass with hyperechoic foci (fat, calcification), shadowing tip-of-iceberg sign.
Complications — torsion (10 to 15 percent), rupture, chemical peritonitis, malignant transformation (1 percent — usually squamous cell carcinoma in postmenopausal women), infection.
Treatment — laparoscopic cystectomy preserving ovary in young women.
Brenner tumour
Pathology — rare; epithelial tumour with transitional-cell (urothelial-like) nests in fibrous stroma. Usually small, solid, unilateral, benign.
Borderline tumors (tumors of low malignant potential)
Pathology — epithelial proliferation and atypia without stromal invasion. Serous and mucinous subtypes.
Behaviour — indolent; 90 percent five-year survival even at advanced stage.
Treatment — surgical staging plus oophorectomy. Conservative fertility-sparing surgery (unilateral salpingo-oophorectomy) in young women with stage I. No adjuvant chemotherapy.
Malignant tumors
Epithelial ovarian carcinoma (90 percent of malignancies)
Subtypes
- High-grade serous carcinoma (HGSC) — most common (70 percent); BRCA1/2 mutations in 15 to 20 percent; p53 mutations universal. Fallopian-tube origin (STIC lesions) increasingly accepted.
- Low-grade serous carcinoma — KRAS/BRAF mutations; relatively chemoresistant; better prognosis.
- Mucinous carcinoma — must exclude GI primary; KRAS mutations.
- Endometrioid carcinoma — associated with endometriosis and Lynch syndrome.
- Clear cell carcinoma — endometriosis-associated; chemoresistant; higher thromboembolism risk.
Risk factors — increasing age, nulliparity, early menarche, late menopause, BRCA1/2 mutation, Lynch syndrome, hormone replacement therapy, endometriosis (clear cell, endometrioid).
Protective — multiparity, breastfeeding, oral contraceptive pill (50 percent reduction after 5 years), tubal ligation, salpingectomy.
Presentation — vague abdominal bloating, early satiety, urinary urgency, pelvic pain. The "ovarian cancer symptom index" includes any of these symptoms more than 12 days per month with onset within the past year — should trigger pelvic USG and CA-125.
Germ cell tumors (5 percent)
Affect young women (under 30 years).
- Dysgerminoma — commonest malignant germ cell; female counterpart of seminoma; bilateral in 10 percent; LDH elevated; very radiosensitive and chemosensitive.
- Endodermal sinus (yolk sac) tumour — Schiller-Duval bodies on histology; AFP elevated; rapidly growing in adolescents.
- Embryonal carcinoma — both AFP and beta-HCG elevated.
- Choriocarcinoma — beta-HCG very high; haemorrhagic, lung metastases.
- Immature teratoma — graded by neuroepithelial content; the only teratoma that is malignant; can secrete AFP.
Treatment — BEP chemotherapy (bleomycin, etoposide, cisplatin); fertility-sparing surgery often feasible.
Sex cord stromal tumors (5 percent)
- Granulosa cell tumour — Call-Exner bodies on histology; inhibin and estradiol secretion (precocious puberty in juvenile type, postmenopausal bleeding and endometrial hyperplasia or carcinoma in adult type — always sample endometrium).
- Theca cell tumour (thecoma) — estrogen-secreting, benign; postmenopausal bleeding.
- Fibroma — benign; Meigs syndrome (ascites, right pleural effusion); resolves with resection.
- Sertoli-Leydig cell tumour — androgen-secreting (virilisation); Reinke crystals.
Metastatic tumors
- Krukenberg tumour — bilateral solid ovarian metastases from a GI primary (most often gastric signet-ring carcinoma); poor prognosis.
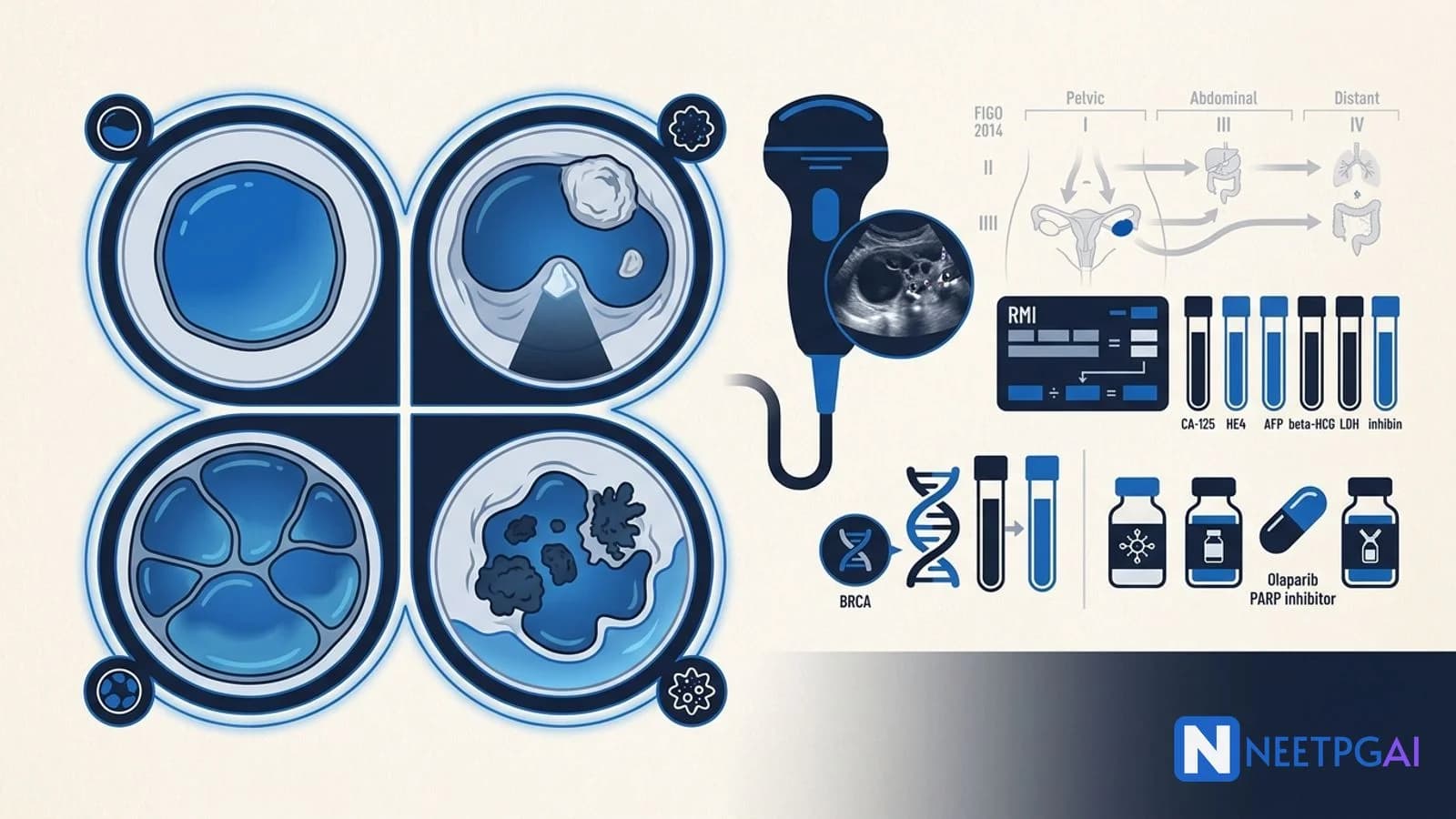
Risk stratification
Tumour markers
| Marker | Tumour |
|---|---|
| CA-125 | Epithelial (esp. serous); also raised in endometriosis, PID, pregnancy, menstruation — limits specificity |
| HE4 | Epithelial; better specificity than CA-125 |
| AFP | Yolk sac, immature teratoma, embryonal |
| Beta-HCG | Choriocarcinoma, embryonal, dysgerminoma (occasional) |
| LDH | Dysgerminoma |
| Inhibin | Granulosa cell |
| CEA | Mucinous (also GI primary) |
| CA 19-9 | Mucinous |
| Estradiol | Granulosa, thecoma |
| Testosterone | Sertoli-Leydig |
Risk of Malignancy Index (RMI)
RMI = Menopausal status × Ultrasound score × CA-125 (IU/mL)
| Component | Score |
|---|---|
| Menopausal status — pre-menopausal | 1 |
| Menopausal status — post-menopausal | 3 |
| Ultrasound score — 0 or 1 feature | 1 |
| Ultrasound score — 2 or more features | 3 |
Ultrasound features: multilocular cyst, solid areas, bilateral, ascites, metastases.
RMI above 200 — refer to gynaecological oncology centre (78 percent sensitivity, 87 percent specificity).
IOTA Simple Rules
International Ovarian Tumor Analysis group offers 5 benign (B) and 5 malignant (M) ultrasound features. If only M features are present, classify as malignant; if only B features, benign; mixed or none, indeterminate (consider MRI or refer).
Surgical staging — FIGO 2014
| Stage | Description |
|---|---|
| I | Limited to ovaries |
| IA | One ovary, capsule intact, no malignant cells in ascites/washings |
| IB | Both ovaries, capsule intact |
| IC | IA or IB with capsule rupture (IC1 — intra-operative; IC2 — pre-operative or surface tumour; IC3 — malignant cells in ascites/washings) |
| II | Pelvic extension |
| IIA | Extension to fallopian tubes / uterus |
| IIB | Extension to other pelvic peritoneal tissues |
| III | Peritoneal spread beyond pelvis or retroperitoneal nodes |
| IIIA1 | Positive retroperitoneal nodes only |
| IIIA2 | Microscopic extra-pelvic peritoneal spread |
| IIIB | Macroscopic peritoneal metastases under 2 cm |
| IIIC | Macroscopic peritoneal metastases above 2 cm (includes liver/spleen capsule) |
| IV | Distant metastases |
| IVA | Malignant pleural effusion (cytology positive) |
| IVB | Parenchymal metastases (liver, spleen, distant nodes, extra-abdominal organs) |
Staging is surgical — exploratory laparotomy with washings, omentectomy, peritoneal biopsies, pelvic and para-aortic lymphadenectomy, total abdominal hysterectomy with bilateral salpingo-oophorectomy.
Treatment
Early-stage (I, IIA)
- Surgery — TAH plus BSO plus comprehensive staging.
- Adjuvant chemotherapy — carboplatin plus paclitaxel for 3 to 6 cycles (stage IC and above; high-grade lesions).
- Fertility-sparing — unilateral salpingo-oophorectomy with comprehensive staging in stage IA grade 1 to 2 disease in young women.
Advanced-stage (III, IV)
- Primary cytoreductive (debulking) surgery — aim for residual disease less than 1 cm (R1) or ideally no visible residual disease (R0).
- Adjuvant chemotherapy — carboplatin plus paclitaxel × 6 cycles.
- Bevacizumab — added in stage III-IV per GOG-218 / ICON7.
- PARP inhibitor maintenance — olaparib (BRCA mutated), niraparib (all-comers), rucaparib. SOLO1, PRIMA, ATHENA-MONO trials.
- Neoadjuvant chemotherapy plus interval debulking — option when primary R0 unlikely (EORTC 55971, CHORUS).
- HIPEC — hyperthermic intraperitoneal chemotherapy after interval debulking, per OVHIPEC trial (modest benefit).
Germ cell tumours
- Surgery — unilateral salpingo-oophorectomy plus staging.
- Chemotherapy — BEP × 3 to 4 cycles; cure rates above 90 percent.
Sex cord stromal
- Surgery — staging plus removal; consider sampling endometrium for granulosa cell.
- Adjuvant — selected high-risk; BEP for advanced.
BRCA testing and risk-reducing surgery
All high-grade epithelial ovarian, fallopian tube, and primary peritoneal carcinoma patients should be offered germline BRCA1/2 testing regardless of family history (NCCN 2025, ICMR 2024).
- BRCA1 — 40 to 60 percent lifetime ovarian cancer risk; offer risk-reducing bilateral salpingo-oophorectomy (RR-BSO) at age 35 to 40 after completion of childbearing.
- BRCA2 — 15 to 25 percent lifetime risk; RR-BSO at age 40 to 45.
- Lynch syndrome — 5 to 12 percent ovarian cancer risk; RR-BSO plus hysterectomy at age 40.
Salpingectomy at time of hysterectomy or sterilisation is now offered prophylactically given fallopian-tube origin of HGSC.
NEET PG MCQ traps
- Most common ovarian tumour overall — mature cystic teratoma (in young) or serous cystadenoma (in adults).
- Most common malignant ovarian tumour — serous cystadenocarcinoma.
- Most common germ cell malignancy — dysgerminoma.
- Schiller-Duval bodies — yolk sac tumour, AFP elevated.
- Call-Exner bodies — granulosa cell tumour, inhibin elevated.
- Reinke crystals — Sertoli-Leydig tumour, virilisation.
- Krukenberg tumour — bilateral, signet-ring cells, GI primary (gastric).
- Meigs syndrome — fibroma plus ascites plus right pleural effusion; resolves with surgery.
- Theca lutein cysts — bilateral, with molar pregnancy or OHSS.
- Mucinous tumours — must rule out appendiceal/GI primary; pseudomyxoma peritonei is usually appendiceal.
- Granulosa cell tumour — postmenopausal bleeding; sample the endometrium for hyperplasia/carcinoma.
- OCP protection — at least 5 years of use halves ovarian cancer risk; strongest in BRCA carriers (controversial breast risk).
Recent updates and Indian context
- SOLO1 (2018) — olaparib maintenance after first-line platinum-based chemotherapy in BRCA-mutated advanced ovarian cancer reduced progression by 70 percent.
- PRIMA (2019) — niraparib maintenance benefits all-comers, with greatest effect in HRD-positive tumours.
- NCCN 2025 — BRCA testing for all high-grade epithelial ovarian cancers; HRD testing to guide PARP-inhibitor selection.
- Indian late presentation — Tata Memorial Hospital data show 75 percent of Indian ovarian cancer patients present at stage III-IV due to absence of population-based screening; CA-125 plus TVS screening has not improved mortality (UKCTOCS) and is not recommended for general population.
- PMJAY coverage — covers debulking surgery and platinum-based chemotherapy; PARP inhibitors are expensive (Lynparza about 4 lakhs/month) but Indian generic olaparib (Cipla, Natco) reduced cost by 60 percent in 2024.
- NEET PG / NEXT alignment — high-yield topics are RMI calculation, FIGO 2014 staging, BRCA-related risk reduction, tumour markers, and Krukenberg / Meigs eponyms.
Frequently asked questions
What is the most common ovarian malignancy?
Epithelial ovarian carcinoma accounts for 90 percent of ovarian malignancies. Serous carcinoma is the commonest subtype (70 percent of epithelial), followed by mucinous, endometrioid, clear cell, and Brenner tumours. High-grade serous carcinoma carries BRCA1/2 mutations in 15 to 20 percent of cases. Median age at diagnosis is 63 years globally; presentations in India tend to be 8 to 10 years younger.
What is the Risk of Malignancy Index (RMI)?
RMI is calculated as menopausal status (1 if pre-menopausal, 3 if post-menopausal) multiplied by ultrasound score (1 if 0 to 1 features, 3 if 2 or more features — multilocular, solid areas, bilateral, ascites, metastases) multiplied by serum CA-125 in IU/mL. An RMI above 200 suggests malignancy with 78 percent sensitivity and 87 percent specificity. Such patients require referral to a gynaecological oncology centre.
What is Meigs syndrome?
Meigs syndrome is the triad of a benign ovarian fibroma (sex cord stromal tumour), ascites, and right-sided pleural effusion that resolves completely after tumour resection. Pseudo-Meigs syndrome describes the same triad with other benign or malignant pelvic tumours including mucinous cystadenoma, Brenner tumour, and metastatic ovarian carcinoma — important to distinguish.
When should BRCA testing be offered?
BRCA1/2 germline testing is recommended for all women with high-grade epithelial ovarian, fallopian tube, or primary peritoneal carcinoma regardless of family history (per NCCN 2025 and ICMR 2024). Carriers face 40 to 60 percent lifetime ovarian cancer risk (BRCA1) and 15 to 25 percent (BRCA2). Risk-reducing bilateral salpingo-oophorectomy at age 35 to 40 (BRCA1) or 40 to 45 (BRCA2) after childbearing is recommended.
What is the standard chemotherapy for ovarian cancer?
Carboplatin plus paclitaxel every 3 weeks for 6 cycles is the global standard for adjuvant treatment of epithelial ovarian cancer (stage IC and above) after debulking surgery. PARP inhibitors (olaparib, niraparib) are added as maintenance therapy in BRCA-mutated or homologous recombination deficient tumours. Bevacizumab is added to carboplatin-paclitaxel in advanced stages. Germ cell tumours use BEP (bleomycin, etoposide, cisplatin).
This content is for educational purposes for NEET PG exam preparation. It is not a substitute for professional medical advice, diagnosis, or treatment. Clinical information has been reviewed by qualified medical professionals.
Written by: NEETPGAI Editorial Team Reviewed by: Pending SME Review Last reviewed: May 2026
Ready to put this into practice?
Start practicing NEET PG MCQs with AI-powered explanations.
Start Free PracticeYour Next Step
Related Study Guides
Anxiety Disorders, OCD & PTSD for NEET PG 2026: Diagnosis, SSRI, CBT
Master anxiety, OCD, and PTSD for NEET PG 2026 — DSM-5 criteria, Y-BOCS, SSRI first-line, exposure-response prevention, ECT indications, India NMHS data.
Cardiovascular Physiology and Cardiac Cycle for NEET PG 2026
Master cardiac cycle, Wiggers diagram, ECG basis, heart sounds, preload-afterload, Starling's law, JVP waves, and CO measurement for NEET PG 2026 — high-yield MCQ traps.
Acute Appendicitis Management for NEET PG 2026: Diagnosis & Surgery
Master acute appendicitis for NEET PG 2026 — pathophysiology, Alvarado score, imaging, laparoscopic appendicectomy, antibiotic-only therapy, complications, MCQ traps.
Join our NEET PG community
Daily MCQs, study tips, and topper strategies on Telegram.
Join on Telegram →