Renal Physiology — GFR, Clearance and Tubular Function for NEET PG 2026
Master GFR, autoregulation, tubular reabsorption, countercurrent multiplier, RAAS, ADH, acid-base handling, and diuretic sites for NEET PG 2026 — high-yield MCQ traps.
Quick Answer
Renal physiology contributes 3 to 5 NEET PG questions every paper across Physiology, Pharmacology, Medicine, and Anaesthesia. Lock these:
- GFR — normal 120 mL/min/1.73 m2; inulin is the gold standard.
- Autoregulation — myogenic plus tubuloglomerular feedback over MAP 80 to 180 mm Hg.
- Clearance — inulin, creatinine (overestimates), iohexol; filtration fraction = GFR/RPF = 0.2.
- Proximal tubule — reabsorbs 65 percent of filtered Na, glucose (Tm), bicarbonate, amino acids.
- Loop of Henle — countercurrent multiplier; medulla up to 1200 mOsm/L.
- RAAS — renin → AT-I → AT-II (via lung ACE) → aldosterone, vasoconstriction, ADH.
- Acid-base — proximal HCO3 reabsorption, distal H+ secretion, NH4+ buffering.
Renal physiology is one of NEET PG's stickiest topics — students under-prepare it because it feels abstract, then lose marks on clearance calculations, diuretic site-of-action questions, and acid-base puzzles that are practically free with the right framework. India-specific stems on snake-bite AKI, hot-climate volume depletion, and high CKD prevalence make it essential.
This NEETPGAI deep dive walks through nephron anatomy, GFR and autoregulation, clearance principles, segment-by-segment tubular function, the countercurrent multiplier, RAAS, ADH and aquaporins, acid-base handling, diuretic sites of action, and high-yield MCQ traps. Pair it with the acid-base disorders guide for a complete renal map.
Nephron anatomy
Each kidney has about 1 million nephrons. Two types:
- Cortical nephrons (85 percent) — short loops of Henle in outer medulla.
- Juxtamedullary nephrons (15 percent) — long loops descending deep into the inner medulla; responsible for urinary concentration.
Functional segments — Bowman's capsule → proximal convoluted tubule (PCT) → proximal straight tubule → thin descending limb → thin ascending limb → thick ascending limb (TAL) → distal convoluted tubule (DCT) → connecting tubule → cortical collecting duct → medullary collecting duct.
Juxtaglomerular apparatus — granular (JG) cells of the afferent arteriole (renin source), macula densa of the early DCT (NaCl sensor), and extraglomerular mesangial cells.
Glomerular filtration rate
GFR = Kf × net filtration pressure, where Kf is the filtration coefficient (surface area × hydraulic permeability).
Net filtration pressure (Starling forces) = (P-GC minus P-BC) minus (pi-GC minus pi-BC), where:
- P-GC = glomerular capillary hydrostatic pressure (about 60 mm Hg)
- P-BC = Bowman's capsule hydrostatic pressure (about 15 mm Hg)
- pi-GC = glomerular capillary oncotic pressure (rises from 28 to 35 mm Hg along the capillary)
- pi-BC = Bowman's capsule oncotic pressure (about 0; albumin filtered minimally)
Net filtration pressure approximately 10 to 17 mm Hg.
Normal GFR = 120 mL/min/1.73 m2 (180 L/day, of which 99 percent is reabsorbed).
Autoregulation
GFR is maintained over MAP 80 to 180 mm Hg by two intrarenal mechanisms:
- Myogenic mechanism — afferent arteriole stretch causes reflex vasoconstriction (fast, seconds).
- Tubuloglomerular feedback (TGF) — macula densa senses NaCl delivery; high NaCl → adenosine release → afferent arteriole constriction (slower, minutes).
Pharmacology of GFR
- ACE inhibitors / ARBs — dilate efferent arteriole → lower glomerular pressure → fall in GFR but renoprotection long term.
- NSAIDs — block prostaglandin-mediated afferent dilation → fall in GFR in volume-depleted states (NSAID-AKI).
- Calcineurin inhibitors — afferent vasoconstriction → reduced GFR (tacrolimus, ciclosporin nephrotoxicity).
- Contrast agents — afferent constriction plus tubular toxicity → contrast-induced AKI.
Clearance
Clearance (C-x) = U-x × V / P-x, where U-x is urine concentration, V is urine flow rate, P-x is plasma concentration.
Inulin clearance = GFR (gold standard) — freely filtered, not reabsorbed, not secreted, not metabolised, not synthesised. Practical issue is the need for continuous IV infusion.
Creatinine clearance — practical surrogate. Overestimates GFR by 10 to 15 percent because creatinine is also secreted by the proximal tubule. Cimetidine and trimethoprim block tubular creatinine secretion → raised serum creatinine without true GFR fall.
Iohexol or 51Cr-EDTA clearance — modern gold-standard alternatives; single-injection method.
PAH clearance = renal plasma flow (RPF) — para-aminohippurate is filtered and almost completely secreted (extraction ratio 90 percent). RPF about 600 mL/min in adults.
Renal blood flow (RBF) = RPF / (1 minus hematocrit) ≈ 1100 mL/min (about 20 to 25 percent of cardiac output).
Filtration fraction (FF) = GFR / RPF = 120 / 600 = 0.2 (20 percent).
eGFR equations
- Cockcroft-Gault — age, weight, creatinine, sex. Older, still used for drug dosing.
- CKD-EPI 2021 (race-free) — current standard in clinical practice. Used to stage CKD into G1 (over 90), G2 (60 to 89), G3a (45 to 59), G3b (30 to 44), G4 (15 to 29), G5 (under 15 or on dialysis).
Tubular reabsorption — segment by segment
Proximal convoluted tubule (PCT)
Reabsorbs about 65 percent of filtered Na+, water, Cl-, K+, and bicarbonate. Reabsorbs 100 percent of glucose and amino acids in health. Isotonic reabsorption (water follows solute).
- Glucose reabsorption via SGLT-2 (90 percent, early PCT) and SGLT-1 (10 percent, late PCT). Tm (transport maximum) about 375 mg/min. Glucose appears in urine when plasma glucose exceeds renal threshold (about 180 mg/dL).
- Bicarbonate reabsorption — H+ secreted via Na-H exchanger (NHE3) combines with filtered HCO3- to form H2CO3 → CO2 plus H2O (catalysed by carbonic anhydrase IV on brush border). CO2 diffuses into the cell, rejoins H2O (carbonic anhydrase II) → HCO3- exits basolaterally.
- Acidic and basic drugs — secreted by organic anion (OAT) and cation (OCT) transporters. Probenecid blocks penicillin secretion.
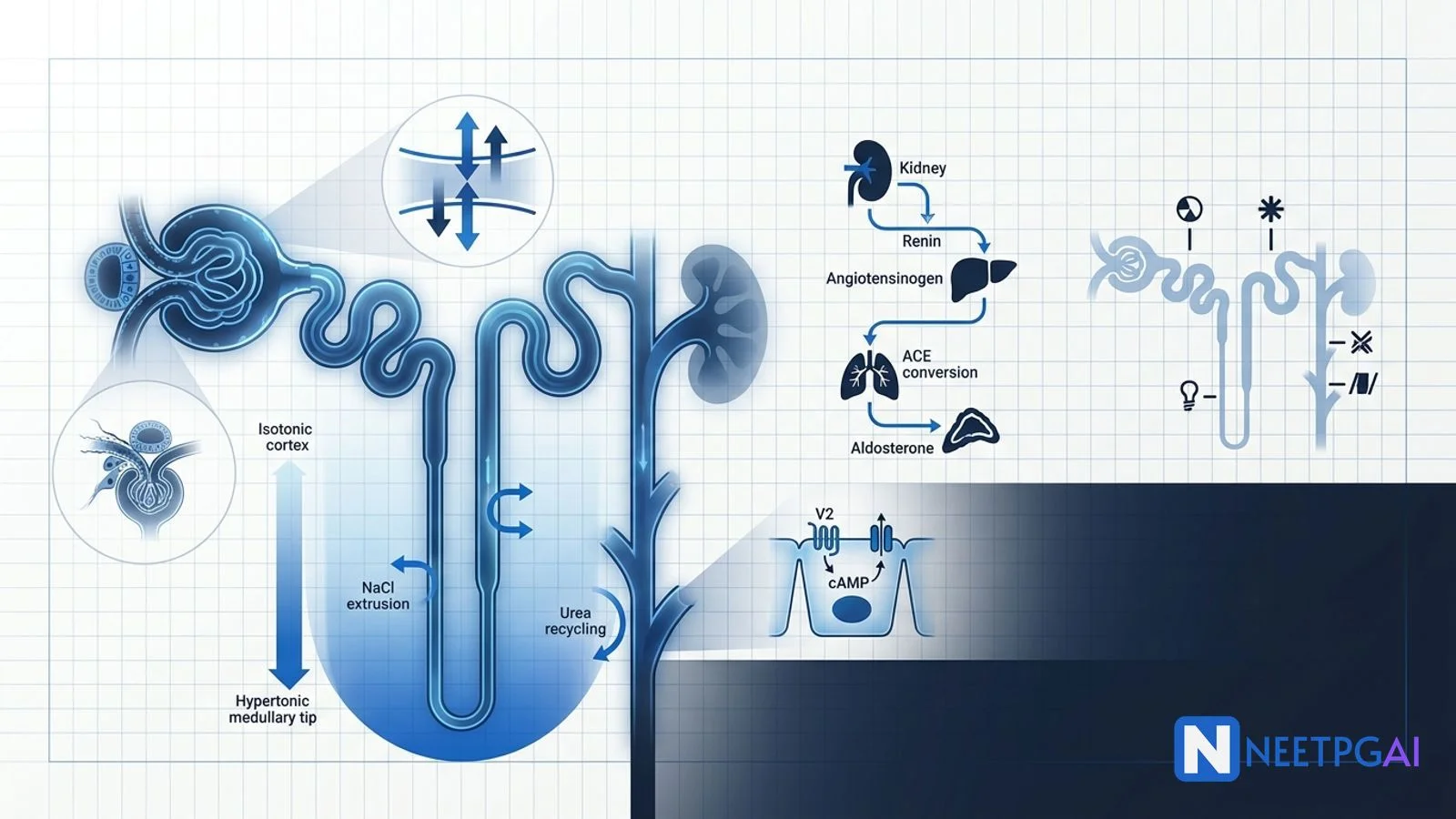
Loop of Henle
The countercurrent multiplier.
- Thin descending limb — permeable to water, impermeable to NaCl. Water exits passively; tubular fluid becomes hypertonic.
- Thin ascending limb — impermeable to water, permeable to NaCl. NaCl exits passively.
- Thick ascending limb — impermeable to water, actively pumps NaCl out via the Na-K-2Cl cotransporter (NKCC2) — site of action of loop diuretics. Reabsorbs about 25 percent of filtered Na+ and is the "diluting segment" — tubular fluid leaves hypotonic (about 100 mOsm/L).
- TAL also reabsorbs Ca2+ and Mg2+ paracellularly (driven by the positive lumen potential from K+ recycling via ROMK).
Distal convoluted tubule (DCT)
- Reabsorbs about 5 percent of filtered Na+ via the Na-Cl cotransporter (NCC) — site of action of thiazides.
- Active Ca2+ reabsorption via TRPV5 channels (PTH-stimulated, thiazide-enhanced).
- Impermeable to water.
Collecting duct (cortical and medullary)
Two cell types in the cortical collecting duct:
- Principal cells — Na+ reabsorbed via ENaC (blocked by amiloride, triamterene); K+ secreted via ROMK (aldosterone-regulated). Site of ADH action on aquaporin-2.
- Intercalated cells — alpha-IC secrete H+ (via H+-ATPase and H+/K+-ATPase) and reabsorb HCO3-; beta-IC secrete HCO3- and reabsorb H+.
Aldosterone
Acts on principal cells — increases Na+ reabsorption and K+/H+ secretion. Released in response to angiotensin II, hyperkalemia, and ACTH (minor).
ADH (vasopressin)
- V1 receptors (vascular) — Gq, vasoconstriction.
- V2 receptors (basolateral, principal cells) — Gs → cAMP → aquaporin-2 insertion into apical membrane → water reabsorption.
Aquaporin-2 is regulated; aquaporin-3 and -4 are constitutive on basolateral side. Loss of AQP2 function or V2 mutation = nephrogenic diabetes insipidus. Lithium and demeclocycline cause acquired nephrogenic DI.
Countercurrent multiplier and urea recycling
The hairpin loop of Henle creates a corticomedullary osmotic gradient (300 mOsm/L cortex to 1200 mOsm/L papillary tip).
- TAL active NaCl extrusion sets up the gradient.
- Vasa recta (countercurrent exchangers) preserve the gradient without washing it out.
- Urea recycling — UT-A1 channels in the inner medullary collecting duct allow ADH-stimulated urea reabsorption; urea is secreted back into the thin descending limb. Contributes about 50 percent of the medullary gradient.
The hypertonic medulla allows ADH-mediated water reabsorption in the collecting duct to concentrate urine to about 1200 mOsm/L. In diabetes insipidus, urine is dilute (under 300 mOsm/L) despite plasma hypertonicity.
RAAS pathway
- Renin — JG cells respond to (a) reduced renal perfusion (afferent baroreceptor), (b) reduced macula densa NaCl, (c) beta-1 sympathetic stimulation.
- Angiotensinogen (from liver) → angiotensin I (decapeptide).
- ACE in pulmonary endothelium → angiotensin II (octapeptide).
- Angiotensin II effects — AT1 receptor: vasoconstriction (efferent more than afferent), aldosterone release, ADH release, thirst, sympathetic activation, cardiac and vascular remodelling.
- Aldosterone from zona glomerulosa — Na+ retention, K+ and H+ excretion.
Negative regulators — atrial natriuretic peptide (ANP) and brain natriuretic peptide (BNP) suppress renin and aldosterone, promote natriuresis.
Acid-base handling
HCO3 reabsorption — proximal tubule
About 4500 mEq/day filtered; 85 to 90 percent reabsorbed in the PCT via the carbonic anhydrase mechanism above; the remainder in the TAL and collecting duct.
H+ secretion — distal nephron
Alpha-intercalated cells secrete H+ via H+-ATPase and H+/K+-ATPase. H+ is buffered in tubular fluid by:
- Filtered HCO3- — minimal in distal nephron (already reabsorbed).
- Phosphate (titratable acid) — about one-third of net acid excretion.
- Ammonia (NH3 / NH4+) — about two-thirds of net acid excretion. Synthesised in PCT from glutamine; NH3 diffuses into the lumen, traps H+ as NH4+. Capacity is highly inducible — rises in chronic acidosis.
Net acid excretion = titratable acid plus NH4+ minus HCO3-. Normally 50 to 100 mEq/day to match endogenous acid production.
Renal tubular acidosis (RTA)
| Type | Defect | Urine pH | Serum K | Clue |
|---|---|---|---|---|
| Type 1 (distal) | Cannot secrete H+ | High (over 5.5) | Low | Calcium stones, nephrocalcinosis |
| Type 2 (proximal) | Cannot reabsorb HCO3 | Low (under 5.5) | Low | Rickets/osteomalacia, Fanconi |
| Type 4 | Aldosterone deficiency/resistance | Low | High | Diabetes, ACEi, NSAIDs |
Diuretic sites of action
| Diuretic class | Site | Mechanism | Key trap |
|---|---|---|---|
| Acetazolamide | PCT | Carbonic anhydrase inhibition | Metabolic acidosis, used in glaucoma, acute mountain sickness |
| Mannitol | Entire nephron (osmotic) | Osmotic diuresis | Contraindicated in anuria, heart failure |
| Loop (furosemide, bumetanide, torsemide) | Thick ascending limb | NKCC2 block | Ototoxicity, hypokalemia, hypocalcemia |
| Thiazides (HCTZ, indapamide, chlorthalidone) | DCT | NCC block | Four hypers (Ca, glucose, urate, lipid) two hypos (Na, K) |
| K-sparing (spironolactone, eplerenone) | Cortical CD | Aldosterone antagonist | Hyperkalemia, gynaecomastia (spironolactone only) |
| K-sparing (amiloride, triamterene) | Cortical CD | ENaC block | Hyperkalemia |
| Tolvaptan, conivaptan | Cortical CD | V2 antagonist | Hyponatremia (SIADH) |
| SGLT-2 inhibitors | PCT | SGLT-2 block | Glycosuria, weight loss, cardio-renal protection |
NEET PG MCQ traps
- Inulin clearance = GFR; creatinine overestimates by 10 to 15 percent due to tubular secretion.
- PAH clearance = RPF; FF = GFR/RPF = 0.2.
- Glucose Tm 375 mg/min; threshold 180 mg/dL.
- TAL is impermeable to water — the "diluting segment".
- Loop diuretics → hypocalcemia; thiazides → hypercalcemia (good for calcium stones).
- Cimetidine and trimethoprim raise serum creatinine without true GFR fall.
- ACEi dilate efferent arteriole → GFR falls but renoprotection long term.
- NSAIDs block prostaglandin-mediated afferent dilation → AKI risk in volume-depleted states.
- ANP and BNP suppress renin and aldosterone; oppose RAAS.
- Aldosterone acts on principal cells (not intercalated).
- ADH acts via V2 on aquaporin-2 in principal cells.
- Urea recycling contributes 50 percent of the medullary gradient.
- Type 1 RTA — distal, urine pH over 5.5, hypokalemia, stones.
- Type 4 RTA — hyperkalemia, low urine pH, common in diabetes.
- Hot-climate volume depletion plus NSAIDs plus ACEi = the classic Indian rural AKI triple-hit on NEET PG vignettes.
Recent updates and Indian context
- NMC CBME 2024 — emphasises mechanism-linked questions on diuretic sites, RAAS pharmacology, and acid-base handling.
- CKD-EPI 2021 (race-free) equation is now the standard eGFR calculator; KDIGO 2024 supports its use.
- SGLT-2 inhibitors — empagliflozin, dapagliflozin, canagliflozin now first-line in diabetic kidney disease and HFrEF irrespective of diabetes status (DAPA-CKD, EMPA-KIDNEY).
- Indian CKD epidemiology — prevalence about 17 percent; major causes are diabetes, hypertension, and CKDu (chronic kidney disease of unknown aetiology) in hot agricultural zones of Andhra Pradesh and Uttar Pradesh.
- Snake-bite AKI — Russell's viper venom causes pigment-induced AKI through rhabdomyolysis and direct nephrotoxicity; classic Indian NEET PG stem.
Frequently asked questions
Why is inulin the gold standard for measuring GFR?
Inulin is a fructose polymer that is freely filtered at the glomerulus and is neither reabsorbed, secreted, metabolised, nor synthesised by the kidney. Its clearance therefore equals GFR exactly. Creatinine clearance overestimates GFR by 10 to 15 percent because creatinine is also secreted by the proximal tubule. Iohexol clearance is the modern practical alternative.
How does tubuloglomerular feedback regulate GFR?
When GFR rises, more NaCl reaches the macula densa cells of the distal convoluted tubule. Macula densa releases adenosine, which constricts the afferent arteriole and reduces GFR back to baseline. NSAIDs blunt this by inhibiting prostaglandin-mediated afferent vasodilation — risky in volume-depleted patients.
What is the countercurrent multiplier and where does it occur?
The countercurrent multiplier is the mechanism by which the loop of Henle generates a hypertonic medullary interstitium (up to 1200 mOsm/L at the papillary tip). The thick ascending limb actively pumps NaCl out (impermeable to water), and the descending limb passively loses water (impermeable to solute). Urea recycling from the inner medullary collecting duct amplifies the gradient.
What stimulates renin release?
Renin is released by juxtaglomerular cells in response to three stimuli — reduced renal perfusion pressure (baroreceptor mechanism in afferent arteriole), reduced NaCl delivery to the macula densa (low intravascular volume), and beta-1 sympathetic stimulation. Renin converts angiotensinogen to angiotensin I; ACE in the lung converts angiotensin I to angiotensin II.
Where do thiazide diuretics act and what are their key side effects?
Thiazides block the Na-Cl cotransporter in the distal convoluted tubule. Key adverse effects — hyponatremia, hypokalemia, hypomagnesemia, hypercalcemia (paradoxical, useful in calcium stones), hyperuricemia (gout), hyperglycemia, and hyperlipidemia. The four hypers and two hypos pattern is a NEET PG favourite.
This content is for educational purposes for NEET PG exam preparation. It is not a substitute for professional medical advice, diagnosis, or treatment. Clinical information has been reviewed by qualified medical professionals.
Written by: NEETPGAI Editorial Team Reviewed by: Pending SME Review Last reviewed: May 2026
Ready to put this into practice?
Start practicing NEET PG MCQs with AI-powered explanations.
Start Free PracticeYour Next Step
Related Study Guides
Anxiety Disorders, OCD & PTSD for NEET PG 2026: Diagnosis, SSRI, CBT
Master anxiety, OCD, and PTSD for NEET PG 2026 — DSM-5 criteria, Y-BOCS, SSRI first-line, exposure-response prevention, ECT indications, India NMHS data.
Carbohydrate Metabolism — Glycolysis, TCA, ETC for NEET PG 2026
Master glycolysis, gluconeogenesis, glycogen metabolism, TCA cycle, ETC, oxidative phosphorylation, PPP, and G6PD deficiency for NEET PG 2026 biochemistry.
Cardiovascular Physiology and Cardiac Cycle for NEET PG 2026
Master cardiac cycle, Wiggers diagram, ECG basis, heart sounds, preload-afterload, Starling's law, JVP waves, and CO measurement for NEET PG 2026 — high-yield MCQ traps.
Join our NEET PG community
Daily MCQs, study tips, and topper strategies on Telegram.
Join on Telegram →